Online Database of Chemicals from Around the World
| Changzhou Tianhua Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tianhuachem.com | |||
 | +86 (519) 8289-1186 | |||
 | +86 (519) 8289-9734 | |||
 | tianhua@tianhuachem.com ctianhua@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1995 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Qingdao Wanyuan Mountain Biontech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bionutrichem.com | |||
 | +86 15092119928 | |||
 | cuiningning@bionutrichem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2018 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Qingdao Truelight Functional Materials Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | en.truelightchem.com | |||
 | +86 13573844393 | |||
 | songjames@truelightchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2008 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Zhengzhou Alfachem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.alfachemch.com | |||
 | +86 (0371) 5505-2911 | |||
 | alfa5@alfachem.cn | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Analytical chemistry >> Standard >> Volatile organic compounds (VOCs) |
|---|---|
| Name | Homosalate |
| Synonyms | 3,3,5-Trimethylcyclohexyl salicylate; Homomenthyl salicylate; Trimethylcyclohenyl salicylate |
| Molecular Structure | 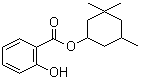 |
| Molecular Formula | C16H22O3 |
| Molecular Weight | 262.35 |
| CAS Registry Number | 118-56-9 |
| EC Number | 204-260-8 |
| SMILES | CC1CC(CC(C1)(C)C)OC(=O)C2=CC=CC=C2O |
| Solubility | <0.1 g/100 mL (water 26 °C), 55mg/mL (DNSO) |
|---|---|
| Density | 1.1±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.539, Calc.* |
| Boiling Point | 341.1±15.0 °C (760 mmHg), Calc.*, 161-165 °C (12 torr) |
| Flash Point | 132.3±13.2 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS07;GHS08;GHS09 Warning Details GHS07;GHS08;GHS09 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335-H361d-H410 Details | ||||||||||||||||||||||||||||
| Safety Statements | P203-P261-P264-P264+P265-P271-P273-P280-P302+P352-P304+P340-P305+P351+P338-P318-P319-P321-P332+P317-P337+P317-P362+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Homosalate, also known as homosalate, is a chemical compound recognized primarily for its role in the formulation of sunscreen products. Its chemical formula is C11H14O3, and it belongs to a class of chemicals known as esters. The compound is derived from salicylic acid, and its synthesis can be traced back to the mid-20th century when the cosmetic industry began to explore more effective UV filters. Homosalate emerged as a promising candidate due to its ability to absorb ultraviolet (UV) radiation, thus protecting the skin from harmful effects. The discovery of homosalate was part of a broader effort to develop safer and more effective sun protection agents. Researchers found that homosalate possesses a broad absorption spectrum, particularly effective in filtering out UVB radiation, which is primarily responsible for sunburn and contributes to skin cancer. As a result, it became a common ingredient in many sunscreen formulations, often used in conjunction with other UV filters to enhance overall protection. In the cosmetic industry, homosalate serves multiple functions beyond UV absorption. It acts as a solvent and a skin conditioning agent, improving the texture and feel of sunscreen products. This versatility makes it a valuable component in the formulation of not only sunscreens but also other personal care products, such as moisturizers and makeup, where sun protection is desired. Homosalate is typically found in concentrations ranging from 10% to 15% in sunscreen formulations. Its efficacy as a UV filter is enhanced when combined with other sunscreen agents, such as octocrylene and avobenzone, creating a synergistic effect that broadens the spectrum of UV protection. Moreover, the stability of homosalate under sunlight makes it a favorable choice for outdoor applications, as it maintains its protective qualities during sun exposure. Despite its widespread use, homosalate has faced scrutiny regarding its safety and environmental impact. Concerns have been raised about the potential for hormone disruption and bioaccumulation in marine ecosystems. Regulatory bodies, including the U.S. Food and Drug Administration (FDA) and the European Commission, have established guidelines for the safe use of homosalate in cosmetic products. These regulations emphasize the importance of conducting safety assessments to evaluate potential health risks associated with its use. In recent years, the demand for natural and organic products has prompted the development of alternative UV filters. However, homosalate remains a popular choice in many conventional formulations due to its effectiveness and stability. Ongoing research continues to explore its properties and potential applications in other areas, such as pharmaceuticals and materials science. In summary, homosalate is a significant chemical compound in the field of sun protection, recognized for its role as a UV filter in sunscreen formulations. Its discovery in the mid-20th century marked a pivotal moment in the cosmetic industry, contributing to advancements in skin safety. While its effectiveness and versatility have cemented its place in personal care products, ongoing research into its safety profile underscores the need for responsible use and development of alternatives in response to consumer preferences. References 2024. Food additive salicylates inhibit human and rat placental 3β-hydroxysteroid dehydrogenase: 3D-QSAR and in silico analysis. Chemico-Biological Interactions. DOI: 10.1016/j.cbi.2024.111203 2023. Single and mixture toxicity evaluation of avobenzone and homosalate to male zebrafish and H295R cells. Chemosphere. DOI: 10.1016/j.chemosphere.2023.140271 2023. Recent developments in tuning the efficacy of different types of sunscreens. Bioprocess and Biosystems Engineering. DOI: 10.1007/s00449-023-02919-9 |
| Market Analysis Reports |