Online Database of Chemicals from Around the World
| Shanghai Yeexin Biochem & Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bioyeexin.com | |||
 | +86 (21) 5417-6788 +86 13671761800 | |||
 | +86 (21) 5417-7952 | |||
 | sales@bioyeexin.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shanghai Yurlic Chemical S&T Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yurlichem.com | |||
 | +86 (21) 37285141 +86 15000957566 | |||
 | +86 (21) 37285142 / 5736-5707 | |||
 | yurlichem@hotmail.com sales@yurlichem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Creative Peptides | USA | |||
|---|---|---|---|---|
 | www.creative-peptides.com | |||
 | +1 (631) 624-4882 | |||
 | +1 (631) 614-7828 | |||
 | info@creative-peptides.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| NetQem, LLC | USA | |||
|---|---|---|---|---|
 | www.netqem.us | |||
 | +1 (919) 544-4122 | |||
 | +1 (919) 544-4109 | |||
 | sales@netqem.us | |||
| Chemical distributor since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Shanghai Hongbang Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hongbangpharma.com | |||
 | +86 (21) 2428-0809 +86 13671516988 | |||
 | +86 (21) 2428-0809 | |||
 | kimberly-hongbangpharma@hotmail.com 1377976036@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Anhui Tokind Advanced Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tokind-mater.com | |||
 | +86 13641793063 | |||
 | sales@tokindchem.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Watanabe Chemical Ind., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.watanabechem.co.jp | |||
 | +81 (82) 231-0540 | |||
 | +81 (82) 231-1451 | |||
 | inquiry@watanabechem.co.jp | |||
| Chemical manufacturer | ||||
| Wanda Group Share Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chinawanda.com | |||
 | +86 (546) 874-5908 | |||
 | +86 (546) 206-1318 | |||
 | wanda@chinawanda.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Common amino acids and protein drugs |
|---|---|
| Name | D-Homoserine |
| Synonyms | (R)-(+)-2-Amino-4-hydroxybutyric acid |
| Molecular Structure | 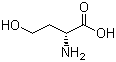 |
| Molecular Formula | C4H9NO3 |
| Molecular Weight | 119.12 |
| Protein Sequence | X |
| CAS Registry Number | 6027-21-0 |
| EC Number | 611-961-6 |
| SMILES | C(CO)[C@H](C(=O)O)N |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 205 °C (Decomposes) (Expl.) |
| Boiling point | 368.7±32.0 °C 760 mmHg (Calc.)* |
| Flash point | 176.8±25.1 °C (Calc.)* |
| Solubility | soluble (water) (Expl.) |
| Index of refraction | 1.511 (Calc.)* |
| Alpha | 9 ° (c=5, h2o) (Expl.), 9 ° (c=5, H2O) (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
D-Homoserine is a non-proteinogenic amino acid and the D-enantiomer of homoserine, which is structurally similar to serine but possesses an additional methylene group in its side chain. It occurs as an intermediate in certain microbial metabolic pathways, particularly in microorganisms that produce D-amino acids for structural or regulatory purposes. While L-homoserine plays a well-known role in the biosynthesis of essential amino acids such as threonine, methionine, and isoleucine, D-homoserine is less common in nature and is often generated through the action of racemases that convert the L-isomer to the D-isomer. The synthesis of D-homoserine can be achieved by several methods. Enzymatic racemization is one common biological route, where L-homoserine racemase catalyzes the conversion of L-homoserine into the D-form. Chemical synthesis typically involves multi-step processes starting from precursors such as aspartic acid, with the chirality introduced via asymmetric synthesis or resolved from a racemic mixture through chiral separation techniques. The purity and enantiomeric excess of D-homoserine are important in research and industrial applications. D-homoserine has applications in biochemical research, particularly in studies of enzyme stereospecificity, microbial metabolism, and stereochemical influences on biological activity. It serves as a substrate or inhibitor in enzymatic assays to investigate the specificity of amino acid-utilizing enzymes. In microbiology, D-homoserine may be incorporated into bacterial cell wall components or act as a signaling molecule in certain species, though its roles are generally less widespread than those of more common D-amino acids such as D-alanine and D-glutamate. Industrial interest in D-homoserine is mainly linked to its potential as a chiral building block for the synthesis of pharmaceuticals and fine chemicals. Its functional groups, including the amino group and hydroxyl group, allow for a range of chemical modifications, making it useful for producing intermediates in stereospecific synthesis pathways. In addition, derivatives of D-homoserine have been investigated for potential bioactive properties. Although not abundant in higher organisms, D-homoserine remains a compound of significance in stereochemical studies, amino acid metabolism research, and synthetic chemistry. Its controlled production, either through enzymatic or chemical methods, continues to support ongoing research into chiral amino acid applications. References 2021. Enzymatic cascade systems for D-amino acid synthesis: progress and perspectives. Systems Microbiology and Biomanufacturing, 1(4). DOI: 10.1007/s43393-021-00037-9 2020. Enantiomeric separation of quorum sensing autoinducer homoserine lactones using GC-MS and LC-MS. Analytical and Bioanalytical Chemistry, 412(12). DOI: 10.1007/s00216-020-02534-7 URL: https://pubmed.ncbi.nlm.nih.gov/32193589 2015. Distribution, industrial applications, and enzymatic synthesis of D-amino acids. Applied Microbiology and Biotechnology, 99(8). DOI: 10.1007/s00253-015-6507-3 URL: https://pubmed.ncbi.nlm.nih.gov/25758960 |
| Market Analysis Reports |