Online Database of Chemicals from Around the World
| Shanghai Qiude Biochemical Engineering Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qiude.com | |||
 | +86 (21) 3728-5021 3728-5029 | |||
 | +86 (21) 5171-4507 | |||
 | webmaster@qiude.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Shanghai Yeexin Biochem & Tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bioyeexin.com | |||
 | +86 (21) 5417-6788 +86 13671761800 | |||
 | +86 (21) 5417-7952 | |||
 | sales@bioyeexin.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shanghai Yurlic Chemical S&T Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yurlichem.com | |||
 | +86 (21) 37285141 +86 15000957566 | |||
 | +86 (21) 37285142 / 5736-5707 | |||
 | yurlichem@hotmail.com sales@yurlichem.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Creative Peptides | USA | |||
|---|---|---|---|---|
 | www.creative-peptides.com | |||
 | +1 (631) 624-4882 | |||
 | +1 (631) 614-7828 | |||
 | info@creative-peptides.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai LingjJing Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.lingjingindustrial.com | |||
 | +86 18964694299 | |||
 | lingjingchem@hotmail.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Shanghai Hongbang Medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hongbangpharma.com | |||
 | +86 (21) 2428-0809 +86 13671516988 | |||
 | +86 (21) 2428-0809 | |||
 | kimberly-hongbangpharma@hotmail.com 1377976036@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Pure Chemistry Scientific Inc | USA | |||
|---|---|---|---|---|
 | www.purechems.com | |||
 | +1 (888) 588-9418 | |||
 | +1 (617) 206-9595 | |||
 | sales@purechems.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Anhui Tokind Advanced Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tokind-mater.com | |||
 | +86 13641793063 | |||
 | sales@tokindchem.com | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Wanda Group Share Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chinawanda.com | |||
 | +86 (546) 874-5908 | |||
 | +86 (546) 206-1318 | |||
 | wanda@chinawanda.com | |||
| Chemical manufacturer | ||||
| Aroz Technologies, LLC | USA | |||
|---|---|---|---|---|
 | www.aroztech.com | |||
 | +1 (877) 818-2125 | |||
 | +1 (877) 818-2129 | |||
 | sales@aroztech.com | |||
| Chemical manufacturer | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Watanabe Chemical Ind., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.watanabechem.co.jp | |||
 | +81 (82) 231-0540 | |||
 | +81 (82) 231-1451 | |||
 | inquiry@watanabechem.co.jp | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Alpha--amino acid |
|---|---|
| Name | L-Homoserine |
| Synonyms | L-2-Amino-4-hydroxybutyric acid |
| Molecular Structure | 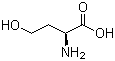 |
| Molecular Formula | C4H9NO3 |
| Molecular Weight | 119.12 |
| Protein Sequence | X |
| CAS Registry Number | 672-15-1 |
| EC Number | 211-590-6 |
| SMILES | C(CO)[C@@H](C(=O)O)N |
| Density | 1.3±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 203 °C (Decomposes) (Expl.) |
| Boiling point | 368.7±32.0 °C 760 mmHg (Calc.)* |
| Flash point | 176.8±25.1 °C (Calc.)* |
| Solubility | water: 1100 g/L (30 °C) (Expl.) |
| Index of refraction | 1.511 (Calc.)* |
| Alpha | -8.5 ° (c=2, H2O 22 °C) (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
L-Homoserine is a non-proteinogenic α-amino acid that serves as an important intermediate in the biosynthetic pathways of several essential amino acids, including threonine, methionine, and isoleucine. It is structurally similar to serine but contains an additional methylene group in its side chain, giving it the molecular formula C4H9NO3. L-Homoserine is found naturally in microorganisms, plants, and some lower eukaryotes but is generally absent from animal proteins. In metabolic pathways such as the aspartate family amino acid pathway, L-homoserine is produced from aspartate via a sequence of enzymatic transformations involving aspartokinase and homoserine dehydrogenase. The discovery of L-homoserine’s role in amino acid biosynthesis emerged from early 20th-century microbial nutrition studies. Researchers observed that certain bacteria and fungi required specific intermediates for growth in minimal media and identified L-homoserine as a critical precursor for threonine and methionine production. This discovery helped establish the metabolic links within the aspartate-derived amino acid family and clarified the role of feedback regulation in amino acid biosynthesis. In nature, L-homoserine is synthesized from L-aspartate through phosphorylation to aspartyl phosphate, reduction to L-aspartate-4-semialdehyde, and subsequent NADPH-dependent reduction to L-homoserine. The molecule’s hydroxyl group allows further enzymatic modifications, such as phosphorylation to O-phospho-L-homoserine, a direct precursor for both threonine and methionine. These reactions occur in the cytosol of plants and microorganisms and are tightly regulated by feedback inhibition to balance amino acid supply. Industrial applications of L-homoserine primarily involve its role as a fermentation product and chemical precursor. Microbial fermentation processes, using organisms such as *Corynebacterium glutamicum*, can be engineered to overproduce L-homoserine for subsequent conversion into other valuable amino acids. Its production is important in the synthesis of L-threonine and L-methionine for use in animal feed, food additives, and pharmaceutical intermediates. L-Homoserine can also serve as a starting material for synthetic routes to various bioactive compounds due to its functional groups, which facilitate further chemical transformations. In biochemical research, L-homoserine is employed as a substrate in studies of amino acid biosynthesis enzymes, metabolic regulation, and stereospecific catalysis. Its availability in both natural and synthetic forms allows controlled experimental investigations of metabolic flux and enzyme specificity. The stability of L-homoserine in aqueous solutions depends on pH and temperature, with the compound prone to slow degradation under strongly acidic or alkaline conditions. In commercial production, purification often involves crystallization or ion-exchange chromatography to achieve the high purity required for biochemical applications. References 2005. Autoinduction in Erwinia amylovora: Evidence of an Acyl-Homoserine Lactone Signal in the Fire Blight Pathogen. Journal of Bacteriology, 187(9). DOI: 10.1128/jb.187.9.3079-3087.2005 URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1082833 1998. Cystathionine gamma-synthase from Arabidopsis thaliana: purification and biochemical characterization of the recombinant enzyme overexpressed in Escherichia coli. The Biochemical Journal, 331(Pt 2). DOI: 10.1042/bj3310639 URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC1219399 1971. The enzymic formation of O-acetylhomoserine in Bacillus subtilis and its regulation by methionine and S-adenosylmethionine. Biochemical and Biophysical Research Communications, 45(5). DOI: 10.1016/0006-291x(71)90478-5 URL: https://pubmed.ncbi.nlm.nih.gov/5001847 |
| Market Analysis Reports |