Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | API >> Synthetic anti-infective drugs >> Antifungal drugs |
|---|---|
| Name | Biapenem |
| Synonyms | 6-[[(4R,5S,6S)-2-Carboxy-6-[(1R)-1-hydroxyethyl]-4-methyl-7-oxo-1-azabicyclo[3.2.0]hept-2-en-3-yl]thio]-6,7-dihydro-5H-pyrazolo[1,2-a][1,2,4]triazol-4-ium inner salt |
| Molecular Structure | 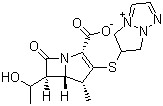 |
| Molecular Formula | C15H18N4O4S |
| Molecular Weight | 350.39 |
| CAS Registry Number | 120410-24-4 (155835-09-9) |
| SMILES | C[C@@H]1[C@@H]2[C@H](C(=O)N2C(=C1SC3CN4C=NC=[N+]4C3)C(=O)[O-])[C@@H](C)O |
| Solubility | >5 mg/mL (H2O, warmed) |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302 Details |
| Safety Statements | P280-P305+P351+P338 Details |
| SDS | Available |
|
Biapenem is a broad-spectrum antibiotic belonging to the class of carbapenems, which are known for their potent bactericidal activity against a wide range of gram-positive and gram-negative bacteria. First discovered in the 1980s, biapenem was developed as part of the ongoing efforts to create new antibiotics that could overcome the growing problem of bacterial resistance to traditional antibiotics. As a carbapenem, biapenem works by inhibiting bacterial cell wall synthesis, a mechanism that is crucial for the bacteria's survival. Its chemical structure features a beta-lactam ring, which is essential for its antibacterial action, and a sulfur atom in the side chain, which distinguishes it from other carbapenems like imipenem. The synthesis of biapenem involves the formation of a beta-lactam ring through a series of chemical reactions, where the precursor compounds are subjected to cyclization reactions in the presence of specific catalysts. The synthesis process has been optimized over the years, with improvements focusing on increasing yield, reducing costs, and ensuring the stability of the final product. Biapenem is typically administered via injection or intravenous infusion in clinical settings, where it is used to treat a variety of infections, including respiratory, urinary tract, and intra-abdominal infections, as well as skin and soft tissue infections. One of the major advantages of biapenem over older antibiotics is its resistance to hydrolysis by many beta-lactamases, enzymes produced by certain bacteria to degrade antibiotics. This makes biapenem an effective option against drug-resistant bacterial strains, including those that produce extended-spectrum beta-lactamases (ESBLs). This feature gives biapenem a significant role in the treatment of infections caused by multi-drug-resistant pathogens, which are becoming an increasing challenge in healthcare settings worldwide. In addition to its antibacterial activity, biapenem has been shown to have a relatively low toxicity profile, making it a safer option for patients compared to some other antibiotics in its class. However, as with all antibiotics, its use must be carefully managed to prevent the development of further resistance. Biapenem is often reserved for severe or hospital-acquired infections, and its use is typically guided by culture and sensitivity testing to ensure it is effective against the specific pathogen causing the infection. Biapenem is not only an important therapeutic agent but also plays a key role in the ongoing research into carbapenem antibiotics. Scientists are continually studying biapenem's mechanisms of action, its effectiveness against emerging resistant strains, and its potential use in combination therapies. Ongoing studies are also exploring the possibility of biapenem being used in the treatment of infections caused by resistant strains of Mycobacterium tuberculosis, as well as its role in treating infections in immunocompromised patients. In summary, biapenem is a vital antibiotic in the fight against bacterial infections, particularly those caused by resistant pathogens. Its discovery and continued development highlight the importance of advancing antibiotic therapies in response to the growing challenge of antimicrobial resistance. References 1990. In vitro activity of LJC10,627, a new carbapenem antibiotic with high stability to dehydropeptidase I. Antimicrobial Agents and Chemotherapy. DOI: 10.1128/aac.34.6.994 2024. Current trend of biapenem susceptibility and disc diffusion breakpoints in Enterobacterales and Pseudomonas aeruginosa. Indian Journal of Medical Microbiology. DOI: 10.1016/j.ijmmb.2024.100695 2024. Determination of Biapenem in Human Blood Plasma by High-Performance Liquid Chromatography with UV Detection. Pharmaceutical Chemistry Journal. DOI: 10.1007/s11094-024-03061-w |
| Market Analysis Reports |