Online Database of Chemicals from Around the World
| MolScanner | Singapore | |||
|---|---|---|---|---|
 | www.molscanner.com | |||
 | +86 18621675448 | |||
 | marketing@molscanner.com | |||
 | WhatsApp:9896 7603 | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Organic raw materials >> Organic fluorine compound >> Fluoropyridine series |
|---|---|
| Name | (3,5-Difluoropyridin-2-yl)methanamine dihydrochloride |
| Molecular Structure | 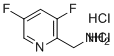 |
| Molecular Formula | C6H8Cl2F2N2 |
| Molecular Weight | 217.04 |
| CAS Registry Number | 1204298-48-5 |
| EC Number | 640-739-1 |
| SMILES | C1=C(C=NC(=C1F)CN)F.Cl.Cl |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H332-H335 Details |
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details |
| SDS | Available |
|
(3,5-Difluoropyridin-2-yl)methanamine dihydrochloride is a chemical compound featuring a pyridine ring substituted with fluorine atoms at the 3- and 5-positions, along with a methanamine group attached to the 2-position of the pyridine ring. The dihydrochloride form consists of two hydrochloride salts bound to the nitrogen atom of the methanamine group. This compound is typically synthesized through methods that selectively introduce the fluorine substituents on the pyridine ring and functionalize the amine group, resulting in a compound with enhanced chemical reactivity. This chemical is primarily used in medicinal chemistry and organic synthesis. The electron-withdrawing fluorine atoms influence the reactivity and stability of the molecule, making it valuable in the design of bioactive molecules. It is particularly useful in the development of drugs, as the difluoropyridine moiety can modulate the electronic properties of the compound, improving its interactions with biological targets such as enzymes and receptors. The methanamine group offers further potential for functionalization, facilitating the creation of compounds with varied biological activity. (3,5-Difluoropyridin-2-yl)methanamine dihydrochloride is also employed in the synthesis of agrochemicals. The pyridine ring, along with the reactive amine group, allows for the preparation of molecules with pesticidal, herbicidal, or fungicidal properties. Its ability to undergo further chemical modifications makes it a valuable intermediate in the production of novel agrochemicals. The biological activity of this compound is influenced by its structure, which combines a fluorinated pyridine ring with a reactive amine group. Similar compounds are often studied for their antimicrobial, anticancer, and anti-inflammatory properties. The fluorine substituents enhance the bioactivity of the molecule, increasing its affinity for various biological targets. As such, this compound has potential applications in the development of targeted therapies for diseases such as cancer, bacterial infections, and other conditions where modulation of biological processes is required. Overall, (3,5-difluoropyridin-2-yl)methanamine dihydrochloride is a versatile compound with significant potential in pharmaceutical and agrochemical development. Its unique combination of a difluoropyridine structure and a reactive amine group makes it an important intermediate for the creation of novel, bioactive molecules. References none |
| Market Analysis Reports |