Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Zehan Biopharma Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zehanbiopharma.com | |||
 | +86 (21) 6135-0663 +86 13052117465 | |||
 | +86 (21) 6135-0662 | |||
 | sales@zehanbiopharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Abmole Bioscience | China | |||
|---|---|---|---|---|
 | www.abmole.cn | |||
 | +86 (21) 5096-7598 | |||
 | +86 (21) 6838-8872 | |||
 | marketing@abmole.cn | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| BeLang Pharma Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.belangpharma.com | |||
 | +86 13885682355 | |||
 | info@belangpharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Shandong Juntai Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.h2opharm.com | |||
 | +86 15106953682 | |||
 | sales01@h2opharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2024 | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| LKT Laboratories, Inc. | USA | |||
|---|---|---|---|---|
 | www.lktlabs.com | |||
 | +1 (888) 558-5227 | |||
 | +1 (651) 644-8357 | |||
 | peacerli@mbolin-lktlabs.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Inhibitor >> Tyrosine protein kinase/signal transducer and transcriptional activator inhibitor (JAK/STAT) >> JAK inhibitor |
|---|---|
| Name | Filgotinib |
| Synonyms | N-[5-[4-[(1,1-Dioxido-4-thiomorpholinyl)methyl]phenyl][1,2,4]triazolo[1,5-a]pyridin-2-yl]cyclopropanecarboxamide |
| Molecular Structure | 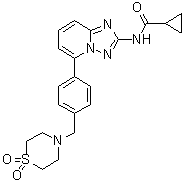 |
| Molecular Formula | C21H23N5O3S |
| Molecular Weight | 425.51 |
| CAS Registry Number | 1206161-97-8 |
| EC Number | 858-411-3 |
| SMILES | C1CC1C(=O)NC2=NN3C(=N2)C=CC=C3C4=CC=C(C=C4)CN5CCS(=O)(=O)CC5 |
| Solubility | 1.8 g/L (water), DMSO |
|---|---|
| Density | 1.51±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.748, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Filgotinib is a novel small molecule drug developed as a selective inhibitor of Janus kinase 1 (JAK1), which plays a crucial role in the signaling pathways of various cytokines involved in inflammatory and immune responses. The discovery of filgotinib is rooted in the need for targeted therapies for autoimmune diseases, particularly rheumatoid arthritis (RA). Filgotinib's development began in the early 2010s, spearheaded by the pharmaceutical company Gilead Sciences in collaboration with Galapagos, a Belgian biotechnology company. The compound was identified through a rigorous drug discovery process that involved screening various chemical entities for their ability to selectively inhibit JAK1 while minimizing off-target effects on other JAK family members. This selectivity is critical, as it enhances the therapeutic potential of the drug while reducing the risk of adverse effects associated with broader JAK inhibition. The synthesis of filgotinib involved a multi-step process, leading to a compound characterized by its specific structure that allows it to effectively bind to the JAK1 enzyme. Filgotinib has shown promising results in clinical trials for several inflammatory conditions. In particular, its application in treating rheumatoid arthritis has garnered significant attention. Clinical studies have demonstrated that filgotinib can significantly reduce the signs and symptoms of RA, improving patient outcomes compared to traditional therapies. The drug's mechanism of action involves inhibiting the JAK1 enzyme, which in turn disrupts the downstream signaling of pro-inflammatory cytokines such as interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α). By doing so, filgotinib effectively mitigates the inflammatory processes that characterize autoimmune diseases. In addition to rheumatoid arthritis, filgotinib is being investigated for its potential use in other autoimmune conditions, including ulcerative colitis and Crohn's disease. The versatility of filgotinib in targeting multiple inflammatory pathways makes it an attractive option for treating various inflammatory disorders. Ongoing clinical trials are exploring the efficacy and safety of filgotinib in these indications, with the aim of providing new therapeutic options for patients who have not responded well to existing treatments. The approval of filgotinib for clinical use marks a significant advancement in the treatment landscape for autoimmune diseases. Its targeted mechanism of action represents a shift towards more personalized medicine, allowing for tailored therapeutic strategies based on individual patient needs. Additionally, filgotinib's favorable safety profile, combined with its efficacy, makes it a valuable addition to the repertoire of available treatments for chronic inflammatory conditions. As research continues, filgotinib may offer new insights into the complex interplay of immune signaling pathways and their role in autoimmune diseases. Its discovery exemplifies the progress being made in the field of targeted therapies, with the potential to improve the quality of life for patients suffering from debilitating conditions. References 2014. Triazolopyridine compounds as selective JAK1 inhibitors: from hit identification to GLPG0634. Journal of Medicinal Chemistry. DOI: 10.1021/jm501262q 2017. Filgotinib (GLPG0634/GS-6034), an oral JAK1 selective inhibitor, is effective in combination with methotrexate (MTX) in patients with active rheumatoid arthritis and insufficient response to MTX: results from a randomised, dose-finding study (DARWIN 1). Annals of the Rheumatic Diseases. DOI: 10.1136/annrheumdis-2016-210104 2024. Real‐World Comparative Effectiveness and Safety of Filgotinib and Upadacitinib for Ulcerative Colitis: A Multicentre Cohort Study. United European Gastroenterology Journal. DOI: 10.1002/ueg2.12704 |
| Market Analysis Reports |