Online Database of Chemicals from Around the World
| BrightGene Bio-medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bright-gene.com | |||
 | +86 (512) 6255-1801 6255-1767 +86 13812696362 | |||
 | +86 (512) 6255-1799 | |||
 | kevinwan@bright-gene.com kevinwan0203@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shuyang Yuqing Pharm Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.yuqingpharm.com | |||
 | +86 (576) 8850-5581 | |||
 | sales@yuqingpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Tecoland Corporation | USA | |||
|---|---|---|---|---|
 | www.tecoland.com | |||
 | +1 (732) 603-9577 | |||
 | +1 (732) 906-1522 | |||
 | info@tecoland.com | |||
| Chemical distributor since 2001 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Biochemical >> Inhibitor >> DNA damage >> DNA/RNA synthesis inhibitor |
|---|---|
| Name | Fidaxomicin |
| Synonyms | R-Tiacumicin B; Tiacumicin B; OPT-80; PAR-101; 3-(((6-Deoxy-4-O-(3,5-dichloro-2-ethyl-4,6-dihydroxybenzoyl)-2-O-methyl-b-D-mannopyranosyl)oxy)-methyl)-12(R)-[(6-deoxy-5-C-methyl-4-O-(2-methyl-1-oxopropyl)-b-D-lyxo-hexopyranosyl)oxy]-11(S)-ethyl-8(S)-hydroxy-18(S)-(1(R)-hydroxyethyl)-9,13,15-trimethyloxacyclooctadeca-3,5,9,13,15-pentaene-2-one; Lipiarmycin |
| Molecular Structure | 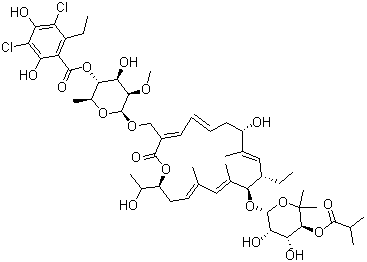 |
| Molecular Formula | 52H74Cl2O18 |
| Molecular Weight | 1058.04 |
| CAS Registry Number | 873857-62-6 |
| EC Number | 692-555-6 |
| SMILES | CC[C@H]1/C=C(/[C@H](C/C=C/C=C(/C(=O)O[C@@H](C/C=C(/C=C(/[C@@H]1O[C@H]2[C@H]([C@H]([C@@H](C(O2)(C)C)OC(=O)C(C)C)O)O)C)C)[C@@H](C)O)CO[C@H]3[C@H]([C@H]([C@@H]([C@H](O3)C)OC(=O)C4=C(C(=C(C(=C4O)Cl)O)Cl)CC)O)OC)O)C |
| Density | 1.3±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.590, Calc.* |
| Boiling Point | 1046.4±65.0 °C (760 mmHg), Calc.* |
| Flash Point | 586.7±34.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302 Details | ||||||||||||
| Safety Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Fidaxomicin is a narrow-spectrum macrocyclic antibiotic that was first discovered in 1975 from *Dactylosporangium aurantiacum*, a soil-dwelling actinomycete. It was initially identified for its potent antibacterial activity, particularly against *Clostridioides difficile*. The discovery of fidaxomicin represented a significant advancement in the treatment of Clostridioides infections, providing a highly selective antimicrobial agent with minimal disruption to the normal gut microbiota. The biosynthesis of fidaxomicin occurs via a polyketide pathway, where enzyme complexes assemble its macrocyclic structure. The unique molecular arrangement of fidaxomicin allows it to inhibit bacterial RNA polymerase by binding to the DNA template-RNA polymerase complex, preventing transcription and subsequent bacterial proliferation. This mode of action is distinct from other antibiotics like rifamycins, contributing to its effectiveness against drug-resistant strains. One of the primary applications of fidaxomicin is in the treatment of *Clostridioides difficile* infections (CDI), where it has demonstrated superior efficacy compared to other antibiotics such as vancomycin. Its narrow-spectrum activity helps preserve beneficial gut bacteria, reducing the recurrence rates of CDI. Fidaxomicin has been widely adopted in clinical settings due to its favorable pharmacokinetics and minimal systemic absorption, which enhances its targeted effect in the gastrointestinal tract. Beyond CDI treatment, fidaxomicin is being investigated for potential applications against other Gram-positive bacterial infections, including *Staphylococcus aureus* and *Enterococcus* species. Research into its broader therapeutic potential continues to explore its role in reducing antimicrobial resistance and improving patient outcomes. Advances in microbial fermentation and synthetic biology are also contributing to improved production efficiency and cost-effectiveness of fidaxomicin. Due to its targeted antimicrobial properties and reduced impact on microbiome diversity, fidaxomicin remains an important antibiotic in managing bacterial infections. Its discovery and continued development highlight the role of natural product-derived antibiotics in modern medicine. References 2024. Potent and specific antibiotic combination therapy against Clostridioides difficile. Nature Chemical Biology, 20(6). DOI: 10.1038/s41589-024-01651-z 2024. Fighting against Clostridioides difficile infection: Current medications. International Journal of Antimicrobial Agents, 64(1). DOI: 10.1016/j.ijantimicag.2024.107198 1983. Structural studies on lipiarmycin I. Characterization by 1H and 13C NMR spectroscopy and isolation of methyl 2-O-methyl-4-O-homodichloroorsellinate-.BETA.-rhamnoside. The Journal of Antibiotics, 36(10). DOI: 10.7164/antibiotics.36.1312 |
| Market Analysis Reports |