Online Database of Chemicals from Around the World
| Richchem International Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chemrg.com | |||
 | +86 (411) 8278-9167 8278-9168 8278-9169 | |||
 | +86 (411) 8807-9241 | |||
 | xie_ruiqiang@chemrg.com | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Shanghai Rokem Industrial Co.Ltd. | China | |||
|---|---|---|---|---|
 | www.rokem.com | |||
 | +86 (21) 5081-1906 +86 18721069645 | |||
 | +86 (21) 5881-8728 | |||
 | milly@rokem.com | |||
| Chemical distributor since 2000 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| LOBA Feinchemie AG | Austria | |||
|---|---|---|---|---|
 | www.loba.co.at | |||
 | +43 (223) 277-391 | |||
 | +43 (223) 276-677 | |||
 | sales@loba.co.at | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2012 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| ProChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.prochemonline.com | |||
 | +1 (815) 398-1788 | |||
 | +1 (815) 398-1810 | |||
 | prochem3@aol.com | |||
| Chemical manufacturer since 1986 | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Whyte Chemicals | UK | |||
|---|---|---|---|---|
 | www.whytechemicals.co.uk | |||
 | +44 (20) 8346-5946 | |||
 | +44 (20) 8349-4589 | |||
 | sales@whytechemicals.co.uk | |||
| Chemical distributor | ||||
| Trigona oHG | Germany | |||
|---|---|---|---|---|
 | www.trigona.de | |||
 | +49 (611) 962-5283 | |||
 | +49 (611) 962-9032 | |||
 | info@trigona.de | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Amino compound >> Cycloalkylamines, aromatic monoamines, aromatic polyamines and derivatives and salts |
|---|---|
| Name | Diphenylamine |
| Synonyms | Diphenylamine redox |
| Molecular Structure | 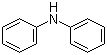 |
| Molecular Formula | C12H11N |
| Molecular Weight | 169.22 |
| CAS Registry Number | 122-39-4 |
| EC Number | 204-539-4 |
| SMILES | C1=CC=C(C=C1)NC2=CC=CC=C2 |
| Density | 1.16 g/mL |
|---|---|
| Melting point | 52-54 °C |
| Boiling point | 302 °C |
| Flash point | 152 °C |
| Water solubility | Slightly soluble. 0.03 g/100 mL |
| Hazard Symbols |     GHS06;GHS07;GHS08;GHS09 Danger Details GHS06;GHS07;GHS08;GHS09 Danger Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H311-H319-H331-H373-H400-H410 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P262-P264-P264+P265-P270-P271-P273-P280-P301+P316-P302+P352-P304+P340-P305+P351+P338-P316-P319-P321-P330-P337+P317-P361+P364-P391-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2811; UN 3077 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Diphenylamine, chemically known as diphenylaminomethane, is an organic compound with the molecular formula C12H11N. It is a member of the amine family, characterized by the presence of two phenyl groups attached to a nitrogen atom. The compound was first synthesized in the 19th century, with its discovery attributed to various researchers who explored the chemistry of anilines and their derivatives. Diphenylamine has gained significant attention due to its versatile applications across multiple industries. The synthesis of diphenylamine typically involves the reaction of aniline with benzene in the presence of an acid catalyst, often utilizing Friedel-Crafts alkylation or amination processes. This reaction results in the formation of diphenylamine along with the release of hydrogen. The compound exhibits a distinctive pale yellow to white crystalline appearance and possesses a characteristic aromatic odor. Diphenylamine is widely used in the rubber industry as an antioxidant. Its ability to prevent the oxidative degradation of rubber products helps to extend their lifespan and maintain their performance. The compound works by scavenging free radicals generated during the oxidation process, thereby protecting the rubber matrix from deterioration. As a result, diphenylamine is commonly found in tires, seals, and various rubber goods, contributing to their durability and resistance to aging. In addition to its role in rubber manufacturing, diphenylamine is employed as a corrosion inhibitor in metalworking fluids. Its ability to form a protective layer on metal surfaces helps to prevent rust and corrosion, making it valuable in the maintenance of machinery and equipment. This application is particularly important in industries such as automotive and aerospace, where metal components are subject to harsh environmental conditions. Diphenylamine also finds use in the production of dyes and pigments. Its chemical structure allows for modifications that lead to the synthesis of various colored compounds, which are then used in textiles, plastics, and coatings. The versatility of diphenylamine derivatives contributes to the development of a wide range of hues and shades, enhancing the aesthetic appeal of consumer products. Moreover, diphenylamine has been utilized in analytical chemistry as a reagent for detecting specific ions. It is often used in the field of food safety, where it serves as an indicator for the presence of nitrite in food products. The reaction of diphenylamine with nitrites results in a color change, providing a visual cue for the presence of these potentially harmful compounds. Despite its numerous applications, diphenylamine is not without concerns regarding its safety and environmental impact. It is classified as a hazardous substance, with potential health effects that may arise from prolonged exposure, including respiratory issues and skin irritation. Regulatory agencies, such as the U.S. Environmental Protection Agency (EPA), have established guidelines to ensure the safe handling and use of diphenylamine in industrial processes. In recent years, there has been a growing emphasis on the development of safer alternatives to diphenylamine, particularly in light of environmental and health considerations. Research efforts are focused on exploring more sustainable practices and finding substitutes that can deliver similar performance without the associated risks. In summary, diphenylamine is a significant compound with diverse applications in the rubber, metalworking, and dye industries. Its discovery and continued use highlight the importance of amines in various chemical processes. As research advances, the focus on safety and sustainability will shape the future of diphenylamine and its derivatives in industrial applications. References 1979. Influences of inducers and inhibitors of the microsomal monooxygenase system on the alkylating intensity of dimethylnitrosamine in mice. Journal of Cancer Research and Clinical Oncology. DOI: 10.1007/bf00405349 1984. Environmental transformations of DPA, SOPP, benomyl, and TBZ. Residue reviews. DOI: 10.1007/978-1-4612-5266-5_4 2024. Preparation and characterization of hydroxyl-terminated polybutadiene graft ferrocene based composite. Journal of Polymer Research. DOI: 10.1007/s10965-024-04204-z |
| Market Analysis Reports |