Online Database of Chemicals from Around the World
| Nanjing Finetech Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fine-chemtech.com | |||
 | +86 (25) 5207-8417 +86 17714198479 | |||
 | +86 (25) 5207-8417 | |||
 | sales@fine-chemtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Taizhou Tongxin Biopharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.allyrise.com | |||
 | +86 18652728585 | |||
 | +86 (523) 8276-5215 | |||
 | sales@allyrise.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Shanghai Rochi Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rochipharma.com | |||
 | +86 (21) 3875-1876 +86 15000076078 | |||
 | +86 (21) 5027-5764 | |||
 | info@rochipharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Nanjing Bic Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bicbiotech.com | |||
 | +86 18851183712 | |||
 | sales@bicbiotech.com | |||
 | QQ Chat | |||
 | WeChat: +8618851183712 | |||
 | WhatsApp:+8618851183712 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | 6-Bromo-4-fluoro-2-methyl-1-(1-methylethyl)-1H-benzimidazole |
| Molecular Structure | 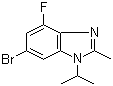 |
| Molecular Formula | C11H12BrFN2 |
| Molecular Weight | 271.13 |
| CAS Registry Number | 1231930-33-8 |
| EC Number | 807-019-0 |
| SMILES | CC1=NC2=C(N1C(C)C)C=C(C=C2F)Br |
| Density | 1.5$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 353.4$+/-$22.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 167.6$+/-$22.3 $degree$C (Calc.)* |
| Index of refraction | 1.601 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H332-H335 Details |
| Safety Statements | P261-P280-P305+P351+P338 Details |
| SDS | Available |
|
6-Bromo-4-fluoro-2-methyl-1-(1-methylethyl)-1H-benzimidazole is a heterocyclic compound belonging to the benzimidazole family, a class of fused bicyclic molecules composed of a benzene ring fused to an imidazole ring. Benzimidazoles have been extensively studied since the early twentieth century for their chemical stability and biological activity, and they have served as important scaffolds in medicinal chemistry, agrochemicals, and materials science. The introduction of substituents such as bromine, fluorine, methyl, and isopropyl groups provides both steric and electronic diversity, which can influence reactivity, pharmacokinetics, and molecular interactions with biological targets. The benzimidazole nucleus was first synthesized in the late 19th and early 20th centuries through condensation reactions of o-phenylenediamines with carboxylic acids or their derivatives. Over time, systematic methods were developed to allow selective functionalization at various positions on the fused ring system. In 6-bromo-4-fluoro-2-methyl-1-(1-methylethyl)-1H-benzimidazole, the bromine at position 6 and the fluorine at position 4 introduce strong electron-withdrawing effects, which can modulate the acidity of the NH group and the overall electron density of the heterocycle. The methyl group at position 2 and the isopropyl substitution at the N1 nitrogen enhance lipophilicity and influence steric interactions, which are important for binding in enzyme active sites or receptor pockets. Compounds of this type have been extensively investigated in pharmaceutical research. Benzimidazole derivatives are well known for their antiviral, antibacterial, antifungal, and antiparasitic activities, and they are also used in the development of proton pump inhibitors, kinase inhibitors, and other therapeutic agents. The presence of halogen substituents, such as bromine and fluorine, is often used to improve metabolic stability, enhance receptor binding affinity, and modulate solubility. The methyl and isopropyl groups further influence pharmacokinetic properties, including absorption and distribution in biological systems. The synthesis of 6-bromo-4-fluoro-2-methyl-1-(1-methylethyl)-1H-benzimidazole typically involves condensation of a suitably substituted o-phenylenediamine with a carboxylic acid or aldehyde derivative under controlled conditions to form the benzimidazole ring. Subsequent selective halogenation and alkylation steps are used to introduce bromine, fluorine, and N-alkyl groups. Modern synthetic methods allow chemists to achieve regioselective substitution and control stereochemistry where relevant, producing high-purity derivatives for further chemical or pharmacological studies. In addition to medicinal applications, substituted benzimidazoles have been explored as intermediates in organic synthesis. The electron-rich heterocyclic ring can participate in cross-coupling reactions, nucleophilic substitutions, and cycloaddition reactions, making these compounds versatile building blocks for the construction of more complex molecular architectures. Functionalization at the halogenated positions allows the introduction of additional substituents or heterocycles, expanding the chemical diversity available for research. The combination of a fused heterocyclic scaffold, halogen substituents, and alkyl groups makes 6-bromo-4-fluoro-2-methyl-1-(1-methylethyl)-1H-benzimidazole a valuable molecule in both synthetic and medicinal chemistry. Its structural features enable detailed studies of structure–activity relationships, receptor binding, and reactivity in heterocyclic chemistry. This compound illustrates how strategic substitution on a benzimidazole core can enhance chemical versatility, improve biological properties, and serve as a platform for further chemical modification in research and drug development. References 2018. Abemaciclib. Pharmaceutical Substances. |
| Market Analysis Reports |