Online Database of Chemicals from Around the World
| Nanjing Finetech Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fine-chemtech.com | |||
 | +86 (25) 5207-8417 +86 17714198479 | |||
 | +86 (25) 5207-8417 | |||
 | sales@fine-chemtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Taizhou Tongxin Biopharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.allyrise.com | |||
 | +86 18652728585 | |||
 | +86 (523) 8276-5215 | |||
 | sales@allyrise.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Nanjing Bic Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bicbiotech.com | |||
 | +86 18851183712 | |||
 | sales@bicbiotech.com | |||
 | QQ Chat | |||
 | WeChat: +8618851183712 | |||
 | WhatsApp:+8618851183712 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Imidazoles |
|---|---|
| Name | 4-Fluoro-2-methyl-1-(1-methylethyl)-6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-benzimidazole |
| Molecular Structure | 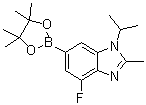 |
| Molecular Formula | C17H24BFN2O2 |
| Molecular Weight | 318.19 |
| CAS Registry Number | 1231930-37-2 |
| EC Number | 813-810-1 |
| SMILES | B1(OC(C(O1)(C)C)(C)C)C2=CC3=C(C(=C2)F)N=C(N3C(C)C)C |
| Density | 1.12$+/-$0.1 g/cm3 (20 $degree$C 760 Torr), Calc.* |
|---|---|
| Boiling point | 442.7$+/-$30.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 221.5$+/-$24.6 $degree$C (Calc.)* |
| Index of refraction | 1.534 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
4-Fluoro-2-methyl-1-(1-methylethyl)-6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-benzimidazole is a functionalized heterocyclic compound belonging to the benzimidazole family, which is composed of a fused benzene and imidazole ring system. Benzimidazoles are widely studied for their chemical stability and pharmacological potential, serving as scaffolds in drug discovery, agrochemicals, and materials science. The molecule is characterized by several substituents: a fluorine atom at position 4, a methyl group at position 2, an isopropyl group at the N1 nitrogen, and a boronate ester group at position 6. Each of these substituents contributes specific electronic, steric, and reactive properties that enhance the compound's utility in chemical synthesis and potential applications in medicinal chemistry. The incorporation of a 4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl group represents the introduction of a boronate ester functionality, which is a widely used organoboron moiety in contemporary synthetic chemistry. Organoboron compounds are known for their stability, ease of handling, and versatility in forming carbon–carbon and carbon–heteroatom bonds. Boronate esters, in particular, are stable under ambient conditions and can participate in transition-metal-catalyzed cross-coupling reactions, such as Suzuki–Miyaura coupling, to construct substituted aromatic and heteroaromatic frameworks. The presence of the boronate ester at the 6-position of the benzimidazole ring allows selective functionalization, enabling chemists to introduce additional substituents or heterocycles while maintaining the integrity of the benzimidazole core. The benzimidazole scaffold itself has been studied extensively in pharmaceutical chemistry because it resembles purine nucleobases and can interact with various biological targets, including enzymes, receptors, and nucleic acid-processing proteins. Substituents such as the fluorine atom, methyl group, and isopropyl group modulate the electronic properties, lipophilicity, and steric profile of the molecule, which can influence binding interactions, metabolic stability, and solubility. These characteristics are particularly valuable in the design of kinase inhibitors, antiviral agents, and other bioactive molecules. Synthetic methods for preparing this compound typically involve constructing the benzimidazole core through condensation of a substituted o-phenylenediamine with a suitable carbonyl compound, followed by selective N-alkylation and halogenation. The boronate ester is commonly introduced via a palladium-catalyzed borylation reaction, allowing regioselective installation of the organoboron group at the desired position on the aromatic ring. These synthetic strategies enable the production of the compound in high purity, facilitating its use in further chemical transformations. The boronate ester functionality provides a highly versatile handle for derivatization. Using cross-coupling reactions, chemists can attach a wide variety of aryl, heteroaryl, or alkyl groups to the benzimidazole framework, creating structurally diverse molecules for chemical and pharmaceutical research. The combination of the boronate ester with the substituted benzimidazole scaffold allows for systematic exploration of structure–activity relationships, including studies on how electronic and steric effects influence reactivity, binding, and biological function. Overall, 4-fluoro-2-methyl-1-(1-methylethyl)-6-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-benzimidazole exemplifies a functionalized heterocyclic compound that bridges synthetic chemistry and medicinal chemistry applications. Its fused heterocyclic core, halogen and alkyl substituents, and reactive boronate ester make it a versatile intermediate for the preparation of more complex molecules and a useful scaffold for exploring chemical reactivity, molecular interactions, and potential biological activity. References 2018. Abemaciclib. Pharmaceutical Substances. |
| Market Analysis Reports |