Online Database of Chemicals from Around the World
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Xinxiang Runyu Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runvmat.com | |||
 | +86 (373) 775-9608 | |||
 | +86 (373) 775-9608 | |||
 | sales@runvmat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | Bis(adamant-1-yl)(2-morpholinophenyl)phosphine |
| Synonyms | 4-(2-(Di(adamantan-1-yl)phosphino)phenyl)morpholine |
| Molecular Structure | 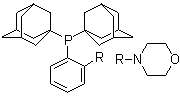 |
| Molecular Formula | C30H42NOP |
| Molecular Weight | 463.63 |
| CAS Registry Number | 1237588-12-3 |
| SMILES | C1COCCN1C2=CC=CC=C2P(C34CC5CC(C3)CC(C5)C4)C67CC8CC(C6)CC(C8)C7 |
| Solubility | Insoluble (1.4E-5 g/L) (25 °C), Calc.* |
|---|---|
| Melting point | 219-224 °C |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2013 ACD/Labs) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P280-P305+P351+P338 Details |
| SDS | Available |
|
Bis(adamant-1-yl)(2-morpholinophenyl)phosphine is a distinctive compound in the field of organophosphorus chemistry, known for its significant role as a ligand in transition metal catalysis. This chemical substance features two adamantyl groups and one 2-morpholinophenyl group attached to a central phosphorus atom. The combination of these groups imparts unique steric and electronic properties that make the compound valuable in various chemical processes. The discovery of Bis(adamant-1-yl)(2-morpholinophenyl)phosphine arose from the need for ligands that provide exceptional steric hindrance and electronic tuning. The adamantyl groups are known for their considerable steric bulk, which can stabilize metal centers and prevent catalyst deactivation by inhibiting aggregation. The 2-morpholinophenyl group introduces both electronic and steric effects due to its structure, which helps modulate the reactivity of the phosphorus center and improve the overall performance of metal catalysts. In practical applications, Bis(adamant-1-yl)(2-morpholinophenyl)phosphine is primarily utilized in transition metal-catalyzed reactions. One of the key areas where this ligand excels is in palladium-catalyzed cross-coupling reactions. The compound's ability to stabilize the palladium center while offering a high degree of control over reaction conditions makes it suitable for various coupling processes, including Suzuki, Heck, and Sonogashira reactions. The steric bulk from the adamantyl groups reduces the tendency of the palladium catalyst to form undesirable by-products, leading to cleaner reactions and higher yields of the desired products. Another notable application is in the field of asymmetric synthesis, where Bis(adamant-1-yl)(2-morpholinophenyl)phosphine is used as a chiral ligand. Its ability to coordinate with transition metals and impart asymmetric induction makes it valuable for the synthesis of enantiomerically pure compounds. This application is particularly important in the pharmaceutical industry, where the precise control over stereochemistry is crucial for developing effective and safe drugs. The compound also finds use in polymerization reactions. Its steric and electronic properties allow it to form stable complexes with various transition metals, which can be employed in the polymerization of olefins and other monomers. This application is beneficial for producing high-performance polymers with specific properties, such as enhanced mechanical strength or thermal stability. Overall, Bis(adamant-1-yl)(2-morpholinophenyl)phosphine represents a significant advancement in ligand design, combining steric and electronic effects to enhance the performance of metal-catalyzed reactions. Its discovery and application underscore the ongoing efforts to improve catalytic processes and develop new materials with tailored properties. References 2011. Rational and Predictable Chemoselective Synthesis of Oligoamines via Buchwald-Hartwig Amination of (Hetero)Aryl Chlorides Employing Mor-DalPhos. The Journal of Organic Chemistry. DOI: 10.1021/jo202358p 2012. Using a P,N-Ligand. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-208-00029 2013. Nonenantioselective Arylation. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-126-00051 |
| Market Analysis Reports |