Online Database of Chemicals from Around the World
| Jinan Chenghui-Shuangda Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jnchsd.com | |||
 | +86 (531) 5889-7051 +86 15053146086 | |||
 | +86 (531) 5889-7093 | |||
 | jnchsd@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Shandong Chengchuang Pharmaceutical Technology Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ccmed.cn | |||
 | +86 (531) 5889-7091 | |||
 | +86 (531) 5889-7092 | |||
 | ccyychem@163.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shandong Chenghui Shuangda Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chsdpharma.com | |||
 | +86-531-58897029 | |||
 | ericqiao@jnchsd.com | |||
 | QQ Chat | |||
 | WeChat: 15550412551 | |||
 | WhatsApp:15550412551 | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2025 | ||||
| ChemBridge Corporation | USA | |||
|---|---|---|---|---|
 | www.chembridge.com | |||
 | +1 (858) 451-7400 | |||
 | +1 (858) 451-7401 | |||
 | sales@chembridge.com | |||
| Chemical manufacturer | ||||
| Scientific Exchange, Inc. | USA | |||
|---|---|---|---|---|
 | www.htscompounds.com | |||
 | +1 (603) 539-7436 | |||
 | +1 (603) 539-7438 | |||
 | sales@htscompounds.com | |||
| Chemical distributor | ||||
| Aronis | Russia | |||
|---|---|---|---|---|
 | www.aronis.ru | |||
 | +7 (926) 578-0336 | |||
 | +7 (495) 426-5336 | |||
 | rakishev@aronis.ru | |||
| Chemical manufacturer | ||||
| MDPI (Molecular Diversity Preservation International) | Switzerland | |||
|---|---|---|---|---|
 | www.mdpi.org | |||
 | +41 (61) 683-7734 | |||
 | +41 (61) 302-8918 | |||
 | samples@mdpi.org | |||
| Chemical manufacturer | ||||
| Advanced Technology & Industrial Co., Ltd. | Hong Kong | |||
|---|---|---|---|---|
 | www.advtechind.com | |||
 | +852 2394-5546 | |||
 | +852 2789-8314 | |||
 | cas@advtechind.com | |||
| Chemical distributor since 1993 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridine compound >> Ethylpyridine |
|---|---|
| Name | Emoxypine succinate |
| Synonyms | Mexidol; 2-ethyl-6-methyl-3-pyridinol succinate |
| Molecular Structure | 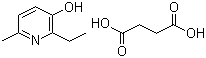 |
| Molecular Formula | C8H11NO.C4H6O4 |
| Molecular Weight | 255.27 |
| CAS Registry Number | 127464-43-1 |
| EC Number | 811-046-3 |
| SMILES | CCC1=C(C=CC(=N1)C)O.C(CC(=O)O)C(=O)O |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Emoxypine succinate is a synthetic antioxidant and membrane-protective agent developed in Russia, primarily used for its neuroprotective, anxiolytic, and antihypoxic effects. Chemically, it is the succinate salt of emoxypine, a 3-hydroxypyridine derivative structurally related to pyridoxine (vitamin B6). The compound combines the biological activity of emoxypine with the metabolic and pharmacological properties of succinic acid, which acts as a Krebs cycle intermediate and participates in cellular energy metabolism. The development of emoxypine and its salts, including emoxypine succinate, occurred during the late 1980s and early 1990s through the work of Russian researchers seeking novel agents that could protect biological membranes and modulate oxidative stress. Emoxypine succinate was introduced under the trade name Mexidol and has since been used clinically in Russia and several other post-Soviet countries. The pharmacological effects of emoxypine succinate are attributed to its antioxidant activity, which includes scavenging of free radicals, inhibition of lipid peroxidation, and enhancement of endogenous antioxidant enzyme activities such as superoxide dismutase. These properties help stabilize cell membranes, improve their fluidity, and protect against damage caused by ischemia, hypoxia, or toxic insults. Emoxypine succinate has been studied in various experimental and clinical settings, including neurology, cardiology, psychiatry, and ophthalmology. In neurology, it has been applied in the treatment of ischemic stroke, traumatic brain injury, and neurodegenerative diseases. Studies have shown that it may help reduce the extent of ischemic damage and improve neurological outcomes by enhancing cerebral blood flow, reducing oxidative stress, and preserving mitochondrial function. In psychiatry, emoxypine succinate has demonstrated anxiolytic and antidepressant-like effects without causing sedation or dependence. Its mechanism is thought to involve modulation of gamma-aminobutyric acid (GABA) and dopamine receptors, as well as normalization of monoamine levels under stress. These properties support its use in treating anxiety disorders, asthenic states, and alcohol withdrawal syndromes. The compound has also been used to manage cardiovascular conditions such as angina pectoris and arrhythmias, where its antioxidant and membrane-stabilizing properties help improve myocardial metabolism and protect cardiac tissues from oxidative damage. In ophthalmology, emoxypine succinate has been investigated for use in degenerative retinal conditions and glaucoma, where it may enhance microcirculation and inhibit free radical-induced retinal injury. Emoxypine succinate is available in both oral and injectable forms. It is typically administered via intramuscular or intravenous injection in hospital settings, and tablets are used for outpatient care. The drug is generally well tolerated, with reported side effects including mild gastrointestinal discomfort, allergic reactions, and hypotension in some individuals. Despite its widespread use in certain regions, emoxypine succinate has not been approved for medical use in the United States, European Union, or Japan. The compound's pharmacological profile has been largely studied in Russian-language literature, and international recognition is limited due to the lack of large-scale, multicenter trials published in peer-reviewed global journals. In summary, emoxypine succinate is a neuroprotective and antioxidant compound developed for clinical use in managing oxidative stress-related conditions, particularly in neurology and psychiatry. Its membrane-stabilizing, antihypoxic, and anxiolytic effects have led to its therapeutic application in several countries, although its use remains regional and is not globally approved. References 2023. Pharmacokinetics of Succinate in Rats after Intravenous Administration of Mexidol. Bulletin of Experimental Biology and Medicine, 175(1). DOI: 10.1007/s10517-023-05810-5 2023. Structure and biological activity of 2-ethyl-3-hydroxy-6-methylpyridinium nitroxysuccinate. Russian Chemical Bulletin, 72(7). DOI: 10.1007/s11172-023-3942-7 2024. The Use of Mexidol in Patients with Mild (Moderate) Cognitive Impairment: Results of a Meta-Analysis. Neuroscience and Behavioral Physiology, 54(5). DOI: 10.1007/s11055-024-01635-5 |
| Market Analysis Reports |