Online Database of Chemicals from Around the World
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Classification | Organic raw materials >> Organometallic compound >> Organic copper |
|---|---|
| Name | (1,10-Phenanthroline)(trifluoromethyl) copper |
| Molecular Structure | 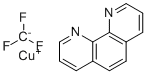 |
| Molecular Formula | C13H8CuF3N2 |
| Molecular Weight | 312.76 |
| CAS Registry Number | 1300746-79-5 |
| EC Number | 696-143-7 |
| SMILES | C1=CC2=C(C3=C(C=CC=N3)C=C2)N=C1.[C-](F)(F)F.[Cu+] |
| Melting point | >230 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319 Details | ||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P280-P301+P317-P302+P352-P305+P351+P338-P317-P321-P330-P332+P317-P337+P317-P362+P364-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
(1,10-Phenanthroline)(trifluoromethyl)copper is an intriguing organometallic compound featuring a copper center coordinated by 1,10-phenanthroline and a trifluoromethyl group. The chemical formula for this compound is typically [Cu(C_13H_8N_2)(CF_3)]^+, where 1,10-phenanthroline acts as a bidentate ligand, coordinating to the copper atom, and trifluoromethyl provides additional properties influencing the compound's reactivity and stability. The discovery of (1,10-Phenanthroline)(trifluoromethyl)copper is a part of ongoing research into organometallic complexes with unique ligand environments. 1,10-Phenanthroline is a well-known chelating agent that binds strongly to metal centers through its nitrogen atoms, forming stable and robust coordination complexes. The addition of the trifluoromethyl group, known for its electron-withdrawing properties, significantly alters the electronic characteristics of the copper center, influencing its reactivity and interaction with substrates. In terms of applications, (1,10-Phenanthroline)(trifluoromethyl)copper has shown significant promise in several areas of chemistry, particularly in catalysis and material science. The trifluoromethyl group imparts unique electronic properties to the complex, making it an effective catalyst for various organic reactions, including C-H activation and cross-coupling reactions. The strong electron-withdrawing nature of the trifluoromethyl group can increase the electrophilicity of the copper center, facilitating reactions that might be less favorable with other substituents. One notable application of this copper complex is in the field of organic synthesis, where it can serve as a catalyst for transformations involving strong C-H bonds. For example, it has been utilized in oxidative coupling reactions and in the formation of carbon-carbon bonds, where its unique ligand environment enhances catalytic efficiency and selectivity. The complex's ability to stabilize reactive intermediates and transition states makes it valuable for producing a variety of organic compounds with high precision. Moreover, (1,10-Phenanthroline)(trifluoromethyl)copper is also of interest in materials science. The properties imparted by the trifluoromethyl group can affect the electronic and optical characteristics of materials in which the copper complex is incorporated. This can lead to the development of novel materials with tailored properties for applications in electronics, photonics, and catalysis. In addition to its practical applications, the study of (1,10-Phenanthroline)(trifluoromethyl)copper contributes to the broader understanding of metal-ligand interactions and the design of new organometallic catalysts. Research into such complexes helps in optimizing reaction conditions, improving catalytic activity, and developing new methodologies for chemical synthesis. In summary, (1,10-Phenanthroline)(trifluoromethyl)copper is a significant organometallic compound with important applications in catalysis and materials science. Its unique ligand environment, combining 1,10-phenanthroline with a trifluoromethyl group, enhances its reactivity and stability, making it a valuable tool in both academic research and practical applications. References 2022. Formation of C(sp)-CF3 Bonds. Science of Synthesis. URL: https://science-of-synthesis.thieme.com/app/text/?id=SD-238-00141 2012. Recent Developments on the Trifluoromethylation of (Hetero)Arenes. Chemistry - An Asian Journal, 7(7). DOI: 10.1002/asia.201200211 |
| Market Analysis Reports |