Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Biochemical >> Biochemical reagent >> Acid-base indicator |
|---|---|
| Name | 1,10-Phenanthroline hydrate |
| Synonyms | o-Phenanthroline monohydrate; 1,10-Phenanthroline monohydrate |
| Molecular Structure | 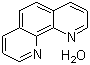 |
| Molecular Formula | C12H8N2.H2O |
| Molecular Weight | 198.22 |
| CAS Registry Number | 5144-89-8 |
| EC Number | 627-114-9 |
| SMILES | C1=CC2=C(C3=C(C=CC=N3)C=C2)N=C1.O |
| Density | 1.10 |
|---|---|
| Melting point | 97-101 °C |
| Water solubility | <0.01 g/100 mL at 21 °C |
| Hazard Symbols |   GHS06;GHS09 Danger Details GHS06;GHS09 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H400-H410 Details | ||||||||||||||||||||||||
| Safety Statements | P264-P270-P273-P301+P316-P321-P330-P405-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
| Transport Information | UN 2811 | ||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||
|
1,10-Phenanthroline hydrate is a coordination compound derived from the well-known heterocyclic organic molecule 1,10-phenanthroline. This compound is distinguished by its ability to form stable chelate complexes with various metal ions, making it a valuable reagent in inorganic and analytical chemistry. The discovery of 1,10-phenanthroline dates back to the early 20th century, when its unique structural properties were first investigated. Since then, it has been utilized extensively in various fields of research and application. The molecular structure of 1,10-phenanthroline features three fused aromatic rings, creating a rigid planar configuration. This geometry allows for effective π-π stacking interactions and the formation of coordination complexes. The hydrate form of this compound contains water molecules that are associated with the 1,10-phenanthroline, influencing its solubility and stability in aqueous environments. The unique properties of 1,10-phenanthroline hydrate arise from its ability to coordinate with transition metals, which significantly enhances its utility in various applications. One of the primary applications of 1,10-phenanthroline hydrate lies in its use as a chelating agent in coordination chemistry. It forms stable complexes with a range of metal ions, including iron, copper, and nickel, making it useful in the study of metal ion behavior and reactivity. These metal-phenanthroline complexes are often employed in analytical techniques such as spectrophotometry and electrochemistry, allowing for the detection and quantification of metal ions in various samples. Additionally, 1,10-phenanthroline hydrate has found applications in the field of biochemistry. Its ability to bind to metal ions has made it a valuable tool in studying metalloproteins and other biological systems that rely on metal ions for their function. For instance, it has been used to investigate the roles of metal ions in enzymatic reactions, as well as to explore the interaction of metal complexes with DNA and other biomolecules. This has implications for understanding mechanisms of drug action and designing metal-based therapeutic agents. Moreover, 1,10-phenanthroline and its metal complexes have garnered attention for their potential use in photophysical applications. The incorporation of metal ions into the phenanthroline framework can lead to unique photophysical properties, making these complexes suitable for use in luminescent materials, sensors, and photovoltaic devices. The ability to tune the properties of these materials through metal coordination opens up new avenues for research in materials science and nanotechnology. In conclusion, 1,10-phenanthroline hydrate represents a versatile and important compound in both coordination chemistry and biochemistry. Its ability to form stable complexes with metal ions has made it an essential tool for researchers in various fields, including analytical chemistry, biochemistry, and materials science. Ongoing studies continue to explore its potential applications, enhancing our understanding of metal interactions in biological systems and paving the way for innovative technologies. References 2024. Integrated application of biochar and chemical fertilizers improves wheat (Triticum aestivum) productivity by enhancing soil microbial activities. Plant and Soil. DOI: 10.1007/s11104-024-06556-3 2023. A Mixed-Ligand Complex of Nickel Trimesinate with 1,10-Phenanthroline as an Adsorbent for Organic Dyes and a Precursor of Nanostructured Materials. Inorganic Materials: Applied Research. DOI: 10.1134/s2075113323020491 2015. Synthesis, DNA interactions and antibacterial PDT of Cu(II) complexes of phenanthroline based photosensitizers via singlet oxygen generation. Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy. DOI: 10.1016/j.saa.2014.11.085 |
| Market Analysis Reports |