Online Database of Chemicals from Around the World
| Sichuan Tongsheng Amino Acids Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.aminoacid.cc | |||
 | +86 (838) 227-4206 250-8248 | |||
 | +86 (838) 227-4207 | |||
 | amino@vip.sina.com sales@biots.cn | |||
 | QQ Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Creative Peptides | USA | |||
|---|---|---|---|---|
 | www.creative-peptides.com | |||
 | +1 (631) 624-4882 | |||
 | +1 (631) 614-7828 | |||
 | info@creative-peptides.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Labseeker Inc | USA | |||
|---|---|---|---|---|
 | www.labseeker.com | |||
 | +1 (858) 750-1632 | |||
 | +1 (858) 412-1220 | |||
 | marketing@labseeker.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shandong Juntai Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.h2opharm.com | |||
 | +86 15106953682 | |||
 | sales01@h2opharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2024 | ||||
| IRIS Biotech GmbH | Germany | |||
|---|---|---|---|---|
 | www.iris-biotech.de | |||
 | +49 (9231) 961-973 | |||
 | +49 (9231) 961-999 | |||
 | info@iris-biotech.de | |||
| Chemical manufacturer since 1788 | ||||
| Ivy Fine Chemicals | USA | |||
|---|---|---|---|---|
 | www.ivychem.com | |||
 | +1 (856) 465-8550 | |||
 | +1 (856) 616-1161 | |||
 | sales@ivychem.com | |||
| Chemical manufacturer | ||||
| Aroz Technologies, LLC | USA | |||
|---|---|---|---|---|
 | www.aroztech.com | |||
 | +1 (877) 818-2125 | |||
 | +1 (877) 818-2129 | |||
 | sales@aroztech.com | |||
| Chemical manufacturer | ||||
| Hamari Chemicals, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.hamarichemicals.com | |||
 | +81 (6) 6322-0191 | |||
 | +81 (6) 6323-2115 | |||
 | sales2@hamari.co.jp | |||
| Chemical manufacturer since 1948 | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Glutamic acid derivative |
|---|---|
| Name | L-Glutamic acid alpha-benzyl ester |
| Synonyms | H-Glu-OBzl |
| Molecular Structure | 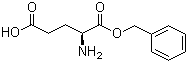 |
| Molecular Formula | C12H15NO4 |
| Molecular Weight | 237.25 |
| Protein Sequence | E |
| CAS Registry Number | 13030-09-6 |
| EC Number | 692-605-7 |
| SMILES | C1=CC=C(C=C1)COC(=O)[C@H](CCC(=O)O)N |
| Melting Point | 157 °C |
|---|---|
| Boiling Point | 416.0±40.0 °C (760 mmHg), Calc.* |
| alpha | 25 |
| Flash Point | 205.4±27.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P305+P351+P338 Details |
| SDS | Available |
|
L-Glutamic acid alpha-benzyl ester, commonly referred to as L-Glu-OBn, is an ester derivative of L-glutamic acid, an important amino acid that plays a crucial role in protein synthesis and metabolic pathways. This compound is characterized by the presence of a benzyl group attached to the alpha-carbon of glutamic acid, which enhances its lipophilicity and influences its biological activity. The discovery of L-glutamic acid alpha-benzyl ester can be attributed to efforts in the early 20th century to explore the properties and applications of amino acid derivatives in various fields, particularly in biochemistry and pharmaceuticals. One of the primary applications of L-glutamic acid alpha-benzyl ester lies in the synthesis of peptide compounds. The benzyl ester group protects the carboxylic acid functionality of glutamic acid during peptide coupling reactions, allowing for the selective formation of peptide bonds without premature deprotection. This property makes L-Glu-OBn a valuable intermediate in the synthesis of peptides and proteins, facilitating the development of biologically active compounds. In addition to its use in peptide synthesis, L-glutamic acid alpha-benzyl ester serves as a key building block in the production of various pharmaceutical agents. Its structural similarity to naturally occurring amino acids enables it to mimic biological processes, making it an attractive candidate for drug design. For example, researchers have utilized L-Glu-OBn in the synthesis of neuroprotective agents and pharmaceuticals targeting the central nervous system, where glutamic acid and its derivatives are known to play vital roles in neurotransmission. Moreover, L-glutamic acid alpha-benzyl ester has potential applications in materials science, particularly in the development of biodegradable polymers and hydrogels. Its incorporation into polymer structures can impart desired mechanical and thermal properties, enhancing the performance of the resulting materials. The ability to modify polymer properties through the inclusion of amino acid derivatives like L-Glu-OBn has led to innovative approaches in the design of biomaterials for drug delivery systems and tissue engineering applications. The biological significance of L-glutamic acid, along with its derivatives, has also sparked interest in exploring its role as a nutritional supplement. L-glutamic acid is recognized for its involvement in metabolic processes and as a precursor for the synthesis of other amino acids and neurotransmitters. Consequently, the esterification of glutamic acid may enhance its stability and bioavailability, promoting its use in dietary supplements aimed at supporting cognitive function and overall health. While the applications of L-glutamic acid alpha-benzyl ester are promising, researchers are also investigating its safety profile and potential toxicity. As with many amino acid derivatives, understanding the metabolic pathways and biological effects of L-Glu-OBn is essential for ensuring its safe use in pharmaceuticals and nutritional products. In summary, L-glutamic acid alpha-benzyl ester is a versatile compound with significant applications in peptide synthesis, pharmaceuticals, materials science, and nutritional supplements. Its discovery and development have contributed to advancements in various fields, underscoring the importance of amino acid derivatives in modern chemistry and biotechnology. References 2018. 1H NMR spectroscopy in the presence of Mosher acid to rapidly determine the enantiomeric composition of amino acid benzyl esters, chirons susceptible to easy racemization. Amino Acids, 50(11). DOI: 10.1007/s00726-018-2653-0 2006. Agaritine gamma-glutamyltransferase. Springer Handbook of Enzymes. DOI: 10.1007/3-540-37717-4_119 1973. Structural Principles. Polymer Chemistry. DOI: 10.1007/978-3-642-65293-6_2 |
| Market Analysis Reports |