Online Database of Chemicals from Around the World
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Taizhou Tongxin Biopharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.allyrise.com | |||
 | +86 18652728585 | |||
 | +86 (523) 8276-5215 | |||
 | sales@allyrise.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shaoxing Fangxiao Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fangxiaochem.com | |||
 | +86 (571) 2829-7060 +86 18969095308 | |||
 | +86 (571) 2829-7065 | |||
 | sales@fangxiaochem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2012 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| East Asia Qifu Pharmaceutical & Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.interchemos.com | |||
 | +86 (25) 835-1527 | |||
 | sales@echemix.com interchemos@163.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Caerulum Pharma Discovery Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.caerulumpharm.com | |||
 | +86 (21) 6455-6180 +86 189149758185 | |||
 | +86 (21) 6455-6180 | |||
 | sales-cpd@caerulumpharma.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Neostar United (Changzhou) Industrial Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.neostarunited.com | |||
 | +86 (519) 8555-7386 +86 18015025600 | |||
 | +86 (519) 8555-7389 | |||
 | marketing1@neostarunited.com | |||
| Chemical distributor since 2014 | ||||
| chemBlink Standard supplier since 2020 | ||||
| E-fine Bio Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.efine-bio.com | |||
 | +86 15251778053 | |||
 | William.efine@hotmail.com | |||
| Chemical manufacturer since 2021 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Classification | API >> Other chemicals |
|---|---|
| Name | Ozanimod |
| Synonyms | 5-[3-[(1S)-2,3-Dihydro-1-[(2-hydroxyethyl)amino]-1H-inden-4-yl]-1,2,4-oxadiazol-5-yl]-2-(1-methylethoxy)benzonitrile |
| Molecular Structure | 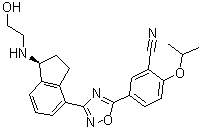 |
| Molecular Formula | C23H24N4O3 |
| Molecular Weight | 404.46 |
| CAS Registry Number | 1306760-87-1 |
| EC Number | 814-246-9 |
| SMILES | CC(C)OC1=C(C=C(C=C1)C2=NC(=NO2)C3=C4CC[C@@H](C4=CC=C3)NCCO)C#N |
| Solubility | Insoluble (1.2E-3 g/L) (25 °C), Calc.*, 10 mM (DMSO) (Expl.) |
|---|---|
| Density | 1.30±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Boiling point | 648.3±65.0 °C 760 mmHg (Calc.)* |
| Flash point | 345.9±34.3 °C (Calc.)* |
| Index of refraction | 1.635 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS08 Danger Details GHS08 Danger Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H372 Details | ||||||||
| Safety Statements | P260-P264-P270-P319-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
| SDS | Available | ||||||||
|
Ozanimod is an oral immunomodulatory drug that selectively targets sphingosine-1-phosphate (S1P) receptor subtypes S1P1 and S1P5. It is primarily used to treat immune-mediated diseases such as relapsing forms of multiple sclerosis (MS) and ulcerative colitis. The compound acts by preventing lymphocytes from exiting lymphoid tissues, thereby reducing their migration to sites of inflammation. This mechanism helps control excessive immune responses while preserving innate immune function. The development of ozanimod followed the clinical success of fingolimod, the first S1P receptor modulator approved for MS. Fingolimod was effective but lacked receptor selectivity, affecting S1P1, S1P3, S1P4, and S1P5, which led to adverse cardiovascular effects such as bradycardia and hypertension. Ozanimod was designed to overcome these limitations by selectively modulating only S1P1 and S1P5, thus minimizing the risk of side effects related to S1P3 activation. Ozanimod was originally developed by Receptos, a biopharmaceutical company focused on autoimmune diseases. In 2015, Celgene acquired Receptos and continued its clinical development. The drug showed high oral bioavailability, predictable pharmacokinetics, and a manageable safety profile in early trials. It was further developed in Phase III clinical trials for relapsing-remitting MS and ulcerative colitis. In 2020, ozanimod received approval from the U.S. Food and Drug Administration (FDA) for the treatment of relapsing forms of MS under the brand name Zeposia. This includes clinically isolated syndrome, relapsing-remitting MS, and active secondary progressive MS. In 2021, it was also approved for the treatment of adults with moderately to severely active ulcerative colitis. The efficacy of ozanimod in MS was demonstrated in two pivotal clinical trials, SUNBEAM and RADIANCE. These trials showed a significant reduction in the annualized relapse rate compared to interferon β-1a. In ulcerative colitis, the TRUENORTH trial showed that ozanimod led to clinical remission, improvement in endoscopic appearance, and sustained clinical response over time. Ozanimod binds selectively to the S1P1 receptor on lymphocytes, leading to receptor internalization and degradation. This action traps lymphocytes in lymph nodes and reduces their presence in peripheral blood, which limits the autoimmune attack on target tissues such as the central nervous system or the colonic mucosa. The drug also acts on S1P5, which is expressed in central nervous system cells like oligodendrocytes, though the clinical significance of this action remains under investigation. The drug is administered once daily and does not require phosphorylation for activation, unlike fingolimod. It also features a step-up dosing regimen during initiation to reduce the risk of bradycardia. Common side effects include nasopharyngitis, headache, and elevated liver enzymes. Serious risks include infections, macular edema, and bradyarrhythmia, but these are considered manageable with monitoring and proper patient selection. Ozanimod represents a second-generation S1P modulator with improved receptor selectivity and safety characteristics. Its development has contributed to the broader understanding of S1P receptor biology and has reinforced the therapeutic value of modulating lymphocyte trafficking in chronic autoimmune conditions. Ongoing studies are evaluating its use in Crohn’s disease and other inflammatory conditions, aiming to expand its clinical utility beyond MS and ulcerative colitis. References 2016. Ozanimod Induction and Maintenance Treatment for Ulcerative Colitis. The New England Journal of Medicine, 374(18). DOI: 10.1056/nejmoa1513248 2024. Ozanimod: A Review in Relapsing Forms of Multiple Sclerosis. CNS Drugs, 38(10). DOI: 10.1007/s40263-024-01116-w 2024. Comparative efficacy of diroximel fumarate, ozanimod and interferon beta-1a for relapsing multiple sclerosis using matching-adjusted indirect comparisons. Journal of Comparative Effectiveness Research, 13(10). DOI: 10.57264/cer-2023-0161 |
| Market Analysis Reports |