Online Database of Chemicals from Around the World
| Jinan Chenghui-Shuangda Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jnchsd.com | |||
 | +86 (531) 5889-7051 +86 15053146086 | |||
 | +86 (531) 5889-7093 | |||
 | jnchsd@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2007 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Genriver Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.genriverpharm.com | |||
 | +86 13761582449 +86 13482015261 | |||
 | +86 (21) 3778-2903 | |||
 | info@genriverpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shandong Chenghui Shuangda Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chsdpharma.com | |||
 | +86-531-58897029 | |||
 | ericqiao@jnchsd.com | |||
 | QQ Chat | |||
 | WeChat: 15550412551 | |||
 | WhatsApp:15550412551 | |||
| Chemical manufacturer since 2014 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | API >> Other chemicals |
|---|---|
| Name | Ozenoxacin |
| Synonyms | 1-Cyclopropyl-8-methyl-7-[5-methyl-6-(methylamino)-3-pyridinyl]-4-oxo-1,4-dihydro-3-quinolinecarboxylic acid; T 3912 |
| Molecular Structure | 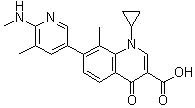 |
| Molecular Formula | C21H21N3O3 |
| Molecular Weight | 363.41 |
| CAS Registry Number | 245765-41-7 |
| SMILES | CC1=CC(=CN=C1NC)C2=C(C3=C(C=C2)C(=O)C(=CN3C4CC4)C(=O)O)C |
| Solubility | Insoluble (1.4E-3 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.372±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Boiling point | 573.5±50.0 °C 760 mmHg (Calc.)* |
| Flash point | 300.7±30.1 °C (Calc.)* |
| Index of refraction | 1.694 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
| SDS | Available |
|
Ozenoxacin is a topical antibacterial agent classified as a non-fluorinated quinolone. It is primarily used for the treatment of impetigo, a common superficial bacterial skin infection mainly caused by Staphylococcus aureus and Streptococcus pyogenes. Ozenoxacin’s mechanism of action involves the inhibition of bacterial DNA gyrase and topoisomerase IV, enzymes essential for bacterial DNA replication, transcription, and repair, resulting in bactericidal activity against susceptible pathogens. Ozenoxacin was developed in the early 21st century as part of efforts to produce effective topical antibiotics with reduced potential for systemic absorption and resistance development. Its chemical structure distinguishes it from fluoroquinolones by lacking a fluorine atom, which is believed to contribute to a more favorable safety profile, particularly reducing risks related to systemic side effects associated with fluoroquinolones. The drug is formulated as a 1% cream for topical application. It is characterized by excellent penetration into the superficial layers of the skin where impetigo lesions occur. Due to its topical application, ozenoxacin achieves high local concentrations at the site of infection with minimal systemic absorption, thereby reducing the likelihood of systemic adverse effects. Clinical trials have demonstrated that ozenoxacin is effective in resolving impetigo lesions, with treatment typically administered twice daily for five days. It exhibits potent activity against methicillin-sensitive and methicillin-resistant Staphylococcus aureus strains, including strains resistant to other topical antibiotics such as mupirocin and fusidic acid, making it a valuable option in cases of antibiotic-resistant skin infections. Ozenoxacin’s safety profile is favorable, with adverse effects limited mostly to mild and transient local reactions such as erythema, pruritus, or burning sensation at the application site. Systemic adverse effects are rare due to low systemic absorption. Its use is contraindicated in patients with known hypersensitivity to quinolones. Pharmacokinetically, after topical application, ozenoxacin demonstrates minimal systemic absorption, with plasma levels often below detectable limits. Metabolism and excretion pathways are not clinically significant due to the limited systemic exposure. In summary, ozenoxacin is a non-fluorinated quinolone topical antibiotic used effectively in the treatment of impetigo. Its targeted mechanism of inhibiting bacterial DNA replication enzymes, combined with minimal systemic absorption and a strong safety profile, supports its clinical utility in managing superficial skin infections, including those caused by resistant bacterial strains. References 2013. In Vitro Activity of Ozenoxacin against Quinolone-Susceptible and Quinolone-Resistant Gram-Positive Bacteria. Antimicrobial Agents and Chemotherapy, 57(12). DOI: 10.1128/AAC.01509-13 2018. Efficacy and Safety of Ozenoxacin Cream for Treatment of Adult and Pediatric Patients With Impetigo: A Randomized Clinical Trial. JAMA Dermatology, 154(7). DOI: 10.1001/jamadermatol.2018.1103 2021. Uptake of Ozenoxacin and Other Quinolones in Gram-Positive Bacteria. International Journal of Molecular Sciences, 22(24). DOI: 10.3390/ijms222413363 |
| Market Analysis Reports |