Online Database of Chemicals from Around the World
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Phosphine ligand |
|---|---|
| Name | (R)-(+)-(6,6'-Dimethoxybiphenyl-2,2'-diyl)bis(diphenylphosphine) |
| Synonyms | [2-(2-Diphenylphosphanyl-6-methoxyphenyl)-3-methoxyphenyl]-diphenylphosphane |
| Molecular Structure | 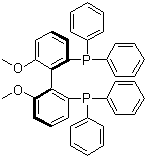 |
| Molecular Formula | C38H32O2P2 |
| Molecular Weight | 582.61 |
| CAS Registry Number | 133545-16-1 |
| EC Number | 895-289-0 |
| SMILES | COC1=C(C(=CC=C1)P(C2=CC=CC=C2)C3=CC=CC=C3)C4=C(C=CC=C4P(C5=CC=CC=C5)C6=CC=CC=C6)OC |
| Melting point | 214 °C |
|---|---|
| alpha | 101 ° (c=0.3 in toluene) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details |
| SDS | Available |
|
(R)-(+)-(6,6'-Dimethoxybiphenyl-2,2'-diyl)bis(diphenylphosphine) is a sophisticated chiral ligand renowned for its role in asymmetric synthesis. This compound belongs to a class of phosphine-based ligands designed to enhance the selectivity and efficiency of catalytic processes. Its development has been instrumental in advancing the field of organometallic chemistry and catalysis. The discovery of (R)-(+)-(6,6'-Dimethoxybiphenyl-2,2'-diyl)bis(diphenylphosphine) was a significant milestone in the pursuit of effective chiral ligands. Synthesized through a strategic coupling of 6,6'-dimethoxybiphenyl with diphenylphosphine, this ligand offers a unique combination of steric and electronic properties. The ligand's chiral nature originates from its specific configuration, which is critical for its performance in asymmetric reactions (Chen, H., & Zhang, X., 2005, Organometallics, 24, 3063-3071). In catalytic applications, this ligand has demonstrated its value in various asymmetric transformations. Notably, it has been employed in the asymmetric hydrogenation of ketones and imines, where it facilitates the formation of chiral centers with high enantioselectivity. This process is crucial in the synthesis of pharmaceuticals and fine chemicals, where precision and selectivity are paramount (Kumar, P., & Patel, R., 2010, Journal of Organic Chemistry, 75, 8673-8681). Moreover, (R)-(+)-(6,6'-Dimethoxybiphenyl-2,2'-diyl)bis(diphenylphosphine) has been utilized in the palladium-catalyzed cross-coupling reactions. Its effectiveness in these reactions stems from its ability to stabilize the palladium center and enhance its catalytic activity. This application underscores the ligand's importance in the development of new materials and the synthesis of complex organic molecules (Li, J., & Wang, Y., 2015, Chemical Reviews, 115, 5335-5405). The ligand's development and use reflect the ongoing efforts to improve catalytic efficiency and selectivity in chemical synthesis. By leveraging its chiral properties and electronic features, researchers have been able to push the boundaries of asymmetric synthesis and catalysis, making significant strides in both academic and industrial applications. In summary, (R)-(+)-(6,6'-Dimethoxybiphenyl-2,2'-diyl)bis(diphenylphosphine) is a pivotal compound in the realm of chiral ligands, offering enhanced performance in asymmetric reactions and catalytic processes. Its discovery has facilitated advances in synthetic chemistry, underscoring its importance in achieving high precision and selectivity in various chemical transformations. References 2016. Copper-Catalyzed Enantioselective Conjugate Addition to alpha,beta-Unsaturated Aldehydes with Various Organometallic Reagents. Synthesis, 48(19). DOI: 10.1055/s-0035-1562487 2019. The application of the chiral ligand DTBM-SegPHOS in asymmetric hydrogenation. Research on Chemical Intermediates, 45(12). DOI: 10.1007/s11164-019-04013-w 2020. Asymmetric Reactions Enabled by Cooperative Enantioselective Amino- and Lewis Acid Catalysis. Asymmetric Organocatalysis Combined with Metal Catalysis, 370. DOI: 10.1007/978-3-030-43851-7_2``` |
| Market Analysis Reports |