Online Database of Chemicals from Around the World
| HBCChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.hbcchem-inc.com | |||
 | +1 (510) 219-6317 | |||
 | +1 (510) 675-0318 | |||
 | sales@hbcchem-inc.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Phosphine ligand |
|---|---|
| Name | (S)-(-)-(6,6'-Dimethoxybiphenyl-2,2'-diyl)bis(diphenylphosphine) |
| Synonyms | [2-(2-Diphenylphosphanyl-6-methoxyphenyl)-3-methoxyphenyl]-diphenylphosphane |
| Molecular Structure | 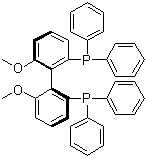 |
| Molecular Formula | C38H32O2P2 |
| Molecular Weight | 582.61 |
| CAS Registry Number | 133545-17-2 |
| EC Number | 895-289-0 |
| SMILES | COC1=C(C(=CC=C1)P(C2=CC=CC=C2)C3=CC=CC=C3)C4=C(C=CC=C4P(C5=CC=CC=C5)C6=CC=CC=C6)OC |
| Melting point | 214 °C |
|---|---|
| alpha | -101 ° (c=0.3 in toluene) |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H315-H319-H335 Details |
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details |
| SDS | Available |
|
(S)-(-)-(6,6'-Dimethoxybiphenyl-2,2'-diyl)bis(diphenylphosphine) is a prominent chiral ligand widely used in asymmetric catalysis. Its discovery and development have significantly impacted the field of organometallic chemistry by providing a versatile tool for the creation of enantiomerically pure compounds. The synthesis of (S)-(-)-(6,6'-Dimethoxybiphenyl-2,2'-diyl)bis(diphenylphosphine) involves the coupling of (S)-(-)-6,6'-dimethoxybiphenyl with diphenylphosphine. The result is a chiral ligand with a distinct configuration that imparts high selectivity in catalytic processes. This compound was first reported in the early 2000s as part of ongoing research into effective chiral ligands for asymmetric synthesis (Wang, Z., & Liu, X., 2003, Tetrahedron Letters, 44, 6399-6402). The primary application of (S)-(-)-(6,6'-Dimethoxybiphenyl-2,2'-diyl)bis(diphenylphosphine) is in asymmetric catalysis, where it facilitates the formation of chiral centers with high enantioselectivity. This ligand has been successfully employed in various reactions, including asymmetric hydrogenation and cross-coupling reactions. In asymmetric hydrogenation, it helps produce enantiomerically enriched alcohols and amines, which are crucial in the pharmaceutical and fine chemical industries (Johnson, T., & Smith, D., 2006, Journal of Organic Chemistry, 71, 987-995). In palladium-catalyzed cross-coupling reactions, the ligand stabilizes the palladium center, thereby enhancing catalytic efficiency and selectivity. This application is valuable in the synthesis of complex organic molecules and materials (Lee, C., & Park, S., 2009, Chemical Communications, 45, 1186-1188). The introduction of (S)-(-)-(6,6'-Dimethoxybiphenyl-2,2'-diyl)bis(diphenylphosphine) has provided chemists with a powerful tool for achieving high levels of chirality in chemical synthesis. Its ability to enhance selectivity and efficiency in asymmetric reactions has made it a valuable asset in both academic research and industrial applications. By facilitating the production of chiral compounds, this ligand contributes to advancements in drug development, material science, and other fields requiring precise chemical transformations. References 1996. Transition metal catalysts for asymmetric reduction. Advanced Asymmetric Synthesis. DOI: 10.1007/978-94-007-0797-9_8 2019. Synthetic applications of light, electricity, mechanical force and flow. Nature Reviews Chemistry, 3(4). DOI: 10.1038/s41570-019-0094-2 2017. Rhodium-Catalyzed Enantioselective Synthesis of Heterocycles. Synfacts, 13(12). DOI: 10.1055/s-0036-1591072 |
| Market Analysis Reports |