Online Database of Chemicals from Around the World
| Gou Hebei Chemicals | India | |||
|---|---|---|---|---|
 | gouhebeichems.store | |||
 | +86 15874272473 | |||
 | info@gouhebeichems.store | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Telly Chemicals Pvt | China | |||
|---|---|---|---|---|
 | tellychemicals.ecer.com | |||
 | +852 52919765 | |||
 | +852 52919765 | |||
 | tellychemicals@protonmail.com | |||
 | Skype Chat | |||
 | WhatsApp:+61 485956223 | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2024 | ||||
| MP Biomedicals LLC. | USA | |||
|---|---|---|---|---|
 | www.mpbio.com | |||
 | +1 (440) 337-1200 | |||
 | +1 (440) 337-1180 | |||
 | inside@mpbio.com | |||
| Chemical manufacturer | ||||
| OChem Incorporation | USA | |||
|---|---|---|---|---|
 | www.ocheminc.com | |||
 | +1 (847) 403-7044 | |||
 | +1 (847) 298-5008 | |||
 | sales@ocheminc.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Amino acids and their derivatives >> Amino alcohol derivative |
|---|---|
| Name | DL-Ephedrine hydrochloride |
| Synonyms | (R*,S*)-(+/-)-alpha-[1-(Methylamino)ethyl]benzyl alcohol hydrochloride |
| Molecular Structure | 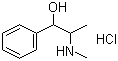 |
| Molecular Formula | C10H15NO.HCl |
| Molecular Weight | 201.69 |
| CAS Registry Number | 134-71-4 |
| EC Number | 205-153-9 |
| SMILES | C[C@H]([C@H](C1=CC=CC=C1)O)NC.Cl |
| Solubility | 10 mM (H2O) |
|---|---|
| Melting point | 188-190 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H317-H412 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P270-P272-P273-P280-P301+P317-P302+P352-P321-P330-P333+P313-P362+P364-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
DL-Ephedrine HCl is an important synthetic variant of ephedrine, a naturally occurring alkaloid derived from the ephedra plant. Recognized for its stimulant and decongestant properties, DL-Ephedrine HCl has played a key role in medicine and various industries since its discovery. Ephedrine was first isolated from ephedra in 1887 by Japanese chemist Nagai Nagayoshi. Synthetic DL-Ephedrine includes both D and L enantiomers, which have a wider range of applications. The hydrochloride form, C10H15NO·HCl, is more stable and more soluble in water, facilitating its use in pharmaceutical preparations. DL-Ephedrine HCl consists of a benzene ring and a double carbon chain containing a hydroxyl (-OH) and an amino (-NH2) group, which are essential for its physiological activity. DL mixtures ensure that the effects of both enantiomers are combined, thereby enhancing its versatile therapeutic efficacy. DL-Ephedrine HCl is widely used as a bronchodilator and decongestant. It is a sympathomimetic that stimulates the sympathetic nervous system by increasing the release of norepinephrine. This results in bronchodilation, making it an effective treatment for asthma, bronchitis, and other respiratory disorders. In addition, its vasoconstrictor properties help relieve nasal congestion. In anesthesia, DL-ephedrine hydrochloride is used to increase blood pressure through vasoconstriction and cardiac stimulation to counteract hypotension during spinal or epidural anesthesia. Due to its central nervous system stimulant effects, it is also used to treat narcolepsy and mild depression. Despite its medical benefits, DL-ephedrine hydrochloride is often abused to enhance athletic performance and lose weight due to its stimulant properties. This has led many countries to introduce regulatory restrictions to limit its supply and prevent abuse. In addition to medicine, DL-ephedrine hydrochloride is used as an organic synthesis precursor for various drugs and fine chemicals. Its stereochemistry provides valuable chiral building blocks for the synthesis of complex molecules. Although DL-ephedrine hydrochloride is safe when used as directed, it has side effects such as increased heart rate, hypertension, and insomnia. Due to its stimulant properties, excessive use can lead to dependence and cardiovascular complications. Regulatory agencies such as the FDA and DEA have implemented strict controls on its sale and distribution to minimize the risks. References 2013. Transport properties of ephedrine hydrochloride through poly(vinyl alcohol) matrices-a simple method for enantiomeric differentiation. Colloid and Polymer Science, 292(8). DOI: 10.1007/s00396-014-3227-7 2015. Wide-range screening of psychoactive substances by FIA-HRMS: identification strategies. Analytical and Bioanalytical Chemistry, 407(15). DOI: 10.1007/s00216-015-8649-7 2016. Enantiomer Separations by Capillary Electrophoresis. Methods in molecular biology (Clifton, N.J.), 1483. DOI: 10.1007/978-1-4939-6403-1_15 |
| Market Analysis Reports |