Online Database of Chemicals from Around the World
| Telly Chemicals Pvt | China | |||
|---|---|---|---|---|
 | tellychemicals.ecer.com | |||
 | +852 52919765 | |||
 | +852 52919765 | |||
 | tellychemicals@protonmail.com | |||
 | Skype Chat | |||
 | WhatsApp:+61 485956223 | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Avonchem/Chromos Express Ltd. | UK | |||
|---|---|---|---|---|
 | www.avonchem.co.uk | |||
 | +44 (1625) 434-300 | |||
 | +44 (1625) 869-777 | |||
 | info@avonchem.co.uk | |||
| Chemical manufacturer | ||||
| Frontier Scientific Services, Inc. | USA | |||
|---|---|---|---|---|
 | www.frontierssi.com | |||
 | +1 (302) 266-6891 (888) 577-2734 | |||
 | +1 (302) 266-8296 | |||
 | customerservice@frontierssi.com | |||
| Chemical manufacturer | ||||
| Classification | API >> Respiratory medication >> Asthma |
|---|---|
| Name | Ephedrine |
| Synonyms | (1R,2S)-(-)-Ephedrine; [R-(R*,S*)]-alpha-[1-(Methylamino)ethyl]-benzenemethanol; L-alpha-(1-Methylaminoethyl)benzyl alcohol |
| Molecular Structure | 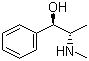 |
| Molecular Formula | C10H15NO |
| Molecular Weight | 165.23 |
| CAS Registry Number | 299-42-3 |
| EC Number | 206-080-5 |
| SMILES | C[C@@H]([C@@H](C1=CC=CC=C1)O)NC |
| Solubility | 63.6 mg/mL (H2O 30 °C) |
|---|---|
| Density | 1.124 |
| Melting point | 37-39 °C |
| Boiling point | 255 °C |
| Flash point | 85 °C |
| alpha | -41 ° (c=5, 1M HCl) |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H314-H317-H318 Details | ||||||||||||||||||||
| Safety Statements | P260-P261-P264-P264+P265-P270-P272-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P330-P333+P317-P362+P364-P363-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Ephedrine is a naturally occurring alkaloid that was first isolated in 1885 by Japanese chemist Nagai Nagayoshi from Ephedra sinica, a plant that has been used in traditional Chinese medicine for thousands of years. Ephedrine has a chemical structure of C10H15NO, consisting of a benzene ring attached to a two-carbon chain with a hydroxyl (-OH) and an amino (-NH2) group. Its structure is similar to amphetamine, which gives it stimulant effects. Ephedrine is primarily used as a bronchodilator and decongestant. It is a sympathomimetic that stimulates the sympathetic nervous system by increasing the release of norepinephrine. This causes bronchodilation, making it an effective treatment for asthma, bronchitis, and other respiratory conditions. In addition, its vasoconstrictive properties help reduce nasal congestion, making it a common ingredient in over-the-counter cold and allergy medications. In anesthesia, ephedrine is used to counteract hypotension (low blood pressure) during spinal or epidural anesthesia. It increases blood pressure through vasoconstriction and stimulates the heart, ensuring adequate blood flow during surgery. Ephedrine is used off-label for its stimulant effects to combat fatigue and improve concentration. However, its use for weight loss and alertness has raised concerns about abuse and adverse effects. Ephedrine works by stimulating adrenergic receptors, specifically alpha and beta receptors. This results in increased heart rate, bronchial dilation, and vasoconstriction. It is able to cross the blood-brain barrier, which also helps stimulate the central nervous system and increase alertness and energy levels. Due to its potential for abuse and side effects, ephedrine is regulated in many countries. In the United States, sales of ephedrine are restricted, and products containing ephedrine are limited to small doses. Long-term or excessive use of ephedrine can lead to adverse effects such as hypertension, palpitations, insomnia, and anxiety. There is also a risk of developing tolerance and dependence. References 1991. Thermogenic synergism between ephedrine and caffeine in healthy volunteers: A double-blind, placebo-controlled study. Metabolism: Clinical and Experimental, 40(3). DOI: 10.1016/0026-0495(91)90117-f 1986. Pharmacological Studies of Two Novel Polycyclic Derivatives of Ephedrine. Archiv der Pharmazie, 319(5). DOI: 10.1002/ardp.19863190506 1971. The Effects of Methoxamine and Ephedrine in Normotensive Pregnant Primates. Anesthesiology, 35(4). DOI: 10.1097/00000542-197110000-00007 |
| Market Analysis Reports |