Online Database of Chemicals from Around the World
| Zhejiang Chempharm Industry&trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chempharm.cn | |||
 | +86 (571) 8770-1568 | |||
 | export@chempharm.cn | |||
| Chemical distributor since 1996 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyridazine |
|---|---|
| Name | Ethyl 4,6-dihydroxypyridazine-3-carboxylate |
| Synonyms | ethyl 4-hydroxy-6-oxo-1H-pyridazine-3-carboxylate |
| Molecular Structure | 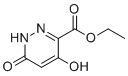 |
| Molecular Formula | C7H8N2O4 |
| Molecular Weight | 184.15 |
| CAS Registry Number | 1352925-63-3 |
| EC Number | 814-463-9 |
| SMILES | CCOC(=O)C1=NNC(=O)C=C1O |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P270-P271-P280-P301+P312-P302+P352-P304+P340-P305+P351+P338-P330-P332+P313-P337+P313-P362-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Ethyl 4,6-dihydroxypyridazine-3-carboxylate is an interesting compound in the field of heterocyclic chemistry, known for its potential applications in pharmaceuticals and organic synthesis. Ethyl 4,6-dihydroxypyridazine-3-carboxylate, with the chemical formula C7H8N2O4, was first synthesized in the mid-20th century during a study focused on exploring novel heterocyclic compounds with potential biological activity. The impetus for this discovery was the need to develop novel scaffolds for drug discovery and to study the reactivity of pyridazine derivatives. The introduction of hydroxyl groups at the 4 and 6 positions of the pyridazine ring and an ethoxylate group at the 3 position resulted in compounds with unique chemical properties and potential applications. The compound is white to pale yellow crystals. It is moderately soluble in common organic solvents such as ethanol and methanol, but has limited solubility in water. The presence of hydroxyl groups enhances its reactivity, making it suitable for a variety of chemical transformations. In addition, the ethoxylate group facilitates its integration into larger molecular structures. Ethyl 4,6-dihydroxypyridazine-3-carboxylate is primarily used in pharmaceutical research as a building block for the synthesis of bioactive molecules. Its unique structure enables it to participate in the formation of a wide range of pharmaceutical compounds, including those with potential anti-inflammatory, antimicrobial, and anticancer properties. The hydroxyl groups provide sites for further chemical modifications, enhancing the versatility of this compound in drug development. In organic synthesis, this compound can serve as an intermediate in the construction of more complex molecules. Its reactivity enables it to undergo a variety of transformations, such as nucleophilic substitution and coupling reactions. These properties make it an important component in the synthesis of heterocyclic compounds, which are widely used in medicinal chemistry and materials science. Recent studies have explored the use of ethyl 4,6-dihydroxypyridine-3-carboxylate in materials science. It has potential applications in the development of new organic materials with unique optical and electronic properties. These materials can be used to make organic semiconductors, light-emitting diodes (LEDs), and other advanced technologies. The compound can also be used as a chemical probe in biochemical research. Its ability to interact with specific biomolecules makes it valuable in studying biological pathways and identifying potential drug targets. Researchers use it to study enzyme activity, protein interactions, and cell signaling mechanisms to gain a deeper understanding of biological processes. Ongoing research continues to discover new applications and improve the efficiency of ethyl 4,6-dihydroxypyridine-3-carboxylate in various fields. Innovations in synthetic methods and materials science are expected to expand its use, providing new opportunities for its application in advanced technologies and medicines. References 2022. Synthesis of Deucravacitinib. Synfacts, 18(8). DOI: 10.1055/s-0041-1738228 |
| Market Analysis Reports |