Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Manus Aktteva Biopharma Llp | China | |||
|---|---|---|---|---|
 | www.manusaktteva.com | |||
 | +91 ((79) 2646-3395 | |||
 | +91 ((79) 2646-3395 | |||
 | staff@manusakttevabiopharma.in | |||
| Chemical distributor since 2010 | ||||
| chemBlink Standard supplier since 2007 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Porphyrin Systems | Germany | |||
|---|---|---|---|---|
 | www.porphyrin-systems.de | |||
 | +49 (40) 6466-5860 | |||
 | +49 (40) 6466-5861 | |||
 | hombrecher@porphyrin-systems.de | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Classification | Analytical chemistry >> Standard >> Pharmacopoeia standards and magazine standards |
|---|---|
| Name | 4-(2-Hydroxyethyl)oxyindole |
| Synonyms | 1,3-Dihydro-4-(2-hydroxyethyl)-2H-indole-2-one |
| Molecular Structure | 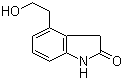 |
| Molecular Formula | C10H11NO2 |
| Molecular Weight | 177.20 |
| CAS Registry Number | 139122-19-3 |
| EC Number | 801-080-7 |
| SMILES | C1C2=C(C=CC=C2NC1=O)CCO |
| Density | 1.3±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.600, Calc.* |
| Boiling Point | 390.0±42.0 °C (760 mmHg), Calc.* |
| Flash Point | 189.7±27.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Danger Details GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H317-H334 Details | ||||||||||||||||||||||||||||
| Safety Statements | P233-P260-P261-P271-P272-P280-P284-P302+P352-P304+P340-P321-P333+P317-P342+P316-P362+P364-P403-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
4-(2-Hydroxyethyl)oxyindole is an organic compound that belongs to the indole class of molecules, characterized by its bicyclic structure consisting of a six-membered benzene ring fused to a five-membered nitrogen-containing pyrrole ring. This particular compound features a hydroxyl group and an ethoxy group attached to the indole core, which gives it distinct chemical properties that have made it a subject of interest in various scientific fields, particularly medicinal chemistry. The discovery of 4-(2-Hydroxyethyl)oxyindole can be traced back to research focused on modifying indole derivatives to enhance their solubility, stability, and biological activity. Indole derivatives, in general, are known for their diverse range of biological activities, including antimicrobial, anti-inflammatory, anticancer, and neuroprotective effects. The addition of a hydroxyl group and an ethoxy group to the indole ring system can potentially influence the compound’s interactions with biological targets, improving its therapeutic potential. The hydroxyl group at the 2-position of the ethoxy group is crucial for enhancing the compound’s polarity and hydrophilicity, which can be beneficial for its bioavailability and transport within the body. The ethoxy group, on the other hand, can impart increased lipophilicity to the molecule, facilitating its ability to cross lipid membranes and enhancing its interactions with cellular components. These modifications may improve the compound's pharmacokinetic properties, making it more suitable for development as a drug candidate. The applications of 4-(2-Hydroxyethyl)oxyindole are diverse, particularly in the realm of drug discovery and development. Its structural features suggest that it may have potential as a lead compound for the development of treatments for neurological disorders, given that indole derivatives have shown promise in modulating neurotransmitter systems and providing neuroprotective effects. The compound could also exhibit antitumor activity, as other indole derivatives have been found to inhibit the growth of cancer cells through various mechanisms, including apoptosis induction and cell cycle disruption. Moreover, 4-(2-Hydroxyethyl)oxyindole may serve as a useful intermediate in the synthesis of other more complex bioactive molecules. Its chemical structure offers opportunities for further modification, allowing researchers to explore a range of derivatives with improved efficacy and selectivity for specific biological targets. By introducing different functional groups or altering the stereochemistry, it may be possible to optimize the compound for various therapeutic applications. In summary, 4-(2-Hydroxyethyl)oxyindole is an intriguing compound that combines the well-established biological activity of indole derivatives with novel modifications aimed at improving drug-like properties. As research progresses, it may contribute to the discovery of new treatments for a variety of diseases, particularly in the fields of cancer and neurology. References 2014. A convenient synthesis of 4-(2-hydroxyethyl)indolin-2-one, a useful intermediate for the preparation of both dopamine receptor agonists and protein kinase inhibitors. Monatshefte für Chemie - Chemical Monthly, 145(7). DOI: 10.1007/s00706-014-1211-z 2013. Synthesis and crystal structure of 1,3-dihydro-4-(2-hydroxyethyl)-2H-indol-2-one. Research on Chemical Intermediates, 40(3). DOI: 10.1007/s11164-013-1368-9 |
| Market Analysis Reports |