Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Shanghai B&C Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bcpharma.com | |||
 | +86 (21) 6597-9781 6597-9782 6597-9783 | |||
 | +86 (21) 6597-9780 | |||
 | info@greenchemintl.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Sichuan Zhongbang Technical Development Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhongbangst.com | |||
 | +86 (830) 258-5019 +86 18982761636 | |||
 | +86 (830) 258-9033 | |||
 | sales@zhongbangst.com sheldon0chen@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shandong Dinghao Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sddhpharm.com | |||
 | +86 (0531) 5856-5868 | |||
 | lily.zhou@sddhpharm.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2018 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Shanghai Qyubiotech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.qyubiotech.com | |||
 | +86 (021) 3759-6619 | |||
 | +86 (021) 3759-6619 | |||
 | lioner@qyubiotech.com | |||
 | QQ Chat | |||
| Chemical distributor since 2015 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Moto Chemistry Int'l Ltd. | China | |||
|---|---|---|---|---|
 | www.motochem.com | |||
 | +86 (756) 322-8461 322-8462 322-8463 | |||
 | +86 (756) 322-8460 | |||
 | moto@motochem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty alcohol |
|---|---|
| Name | 2-Ethyl-2-adamantanol |
| Synonyms | 2-Ethyl-2-hydroxytricyclo[3,3,1,1(3,7)])decane |
| Molecular Structure | 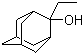 |
| Molecular Formula | C12H20O |
| Molecular Weight | 180.28 |
| CAS Registry Number | 14648-57-8 |
| SMILES | CCC1(C2CC3CC(C2)CC1C3)O |
| Density | 1.0±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.527, Calc.* |
| Boiling Point | 265.5±8.0 °C (760 mmHg), Calc.* |
| Flash Point | 110.2±10.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302 Details |
| Safety Statements | P280-P305+P351+P338 Details |
| SDS | Available |
|
2-Ethyl-2-adamantanol is an organic compound belonging to the adamantane family, which is characterized by a rigid, three-dimensional structure. This compound is a hydroxylated derivative of adamantane, with the hydroxyl group (-OH) attached to the second carbon of an ethyl-substituted adamantane ring structure. The molecular formula for 2-Ethyl-2-adamantanol is C12H18O, and its structure consists of a bicyclic adamantane core with an ethyl group at the second position, making it a member of the class of secondary alcohols. The discovery of adamantane derivatives like 2-ethyl-2-adamantanol dates back to the early 20th century, with the first synthesis of adamantane itself reported in the 1930s. Adamantane and its derivatives, including 2-ethyl-2-adamantanol, were initially studied for their potential use in the pharmaceutical industry due to their unique structural properties. These compounds are of interest in medicinal chemistry due to their stability, which is a result of the rigid, three-dimensional nature of the adamantane structure. The compound 2-ethyl-2-adamantanol has been found to have several practical applications. One of its key uses is as a building block in organic synthesis, particularly in the development of pharmaceuticals and other specialty chemicals. It serves as an intermediate in the preparation of more complex organic molecules due to its functional group, which is capable of undergoing various chemical reactions such as esterification, reduction, or substitution. In addition, 2-ethyl-2-adamantanol has shown potential in the synthesis of fragrances and flavor compounds. The unique adamantane framework can be modified to produce molecules with desirable aromatic properties, and thus 2-ethyl-2-adamantanol and its derivatives are explored for use in the formulation of scents and flavors. The stability and volatility of adamantane-based compounds also make them suitable candidates for incorporation into perfumes, colognes, and other consumer products. Another significant area of application for 2-ethyl-2-adamantanol is in the field of materials science. The rigid structure of adamantane derivatives, including 2-ethyl-2-adamantanol, imparts high thermal stability to materials that incorporate these compounds. This characteristic makes them valuable in the synthesis of polymers and coatings that require enhanced thermal resistance and mechanical strength. These properties are particularly useful in the development of materials used in harsh environments, such as high-performance coatings and advanced polymer composites. While the chemical is not widely known for its pharmaceutical uses, the adamantane core structure itself has been of interest in the development of antiviral drugs, particularly in relation to compounds like amantadine, which has been used to treat influenza. However, the specific applications of 2-ethyl-2-adamantanol in the medical field remain limited, with the compound being more significant in industrial and synthetic chemistry applications. In conclusion, 2-ethyl-2-adamantanol is a functionalized adamantane derivative with important uses in organic synthesis, fragrance production, and materials science. Its stability and versatile chemistry make it a valuable intermediate in the creation of specialty chemicals, and its role in the development of advanced polymers and coatings underscores its importance in materials technology. While it is not as widely known in pharmaceuticals, its chemical structure remains an area of ongoing research, particularly in relation to its potential applications in high-performance materials and organic chemistry. References 2008. Recent Progress in the Catalytic Synthesis of Tertiary Alcohols from Ketones with Organometallic Reagents. Synthesis, 2008(12). DOI: 10.1055/s-2008-1067046 2010. Zinc(II)-Catalyzed Grignard Additions to Ketones with RMgBr and RMgI. Synfacts, 2010(7). DOI: 10.1055/s-0029-1220050 2020. Ligand-Free Nickel-Catalyzed Reductive Allylic Defluorinative Cross-Coupling of α-Trifluoromethyl Alkenes with Epoxides. Synlett, 31(13). DOI: 10.1055/s-0040-1707170 |
| Market Analysis Reports |