Online Database of Chemicals from Around the World
| Futoh Chemicals Co., Ltd. | Japan | |||
|---|---|---|---|---|
 | www.futoh.co.jp | |||
 | +81 (52) 521-8171 | |||
 | +81 (52) 521-8793 | |||
 | tokoro@futoh.co.jp | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2007 | ||||
| Shanghai Sinofluoro chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinofluoro.com | |||
 | +86 (21) 6427-9360 | |||
 | +86 (21) 6427-8603 | |||
 | simon.lv@sinofluoro.com jerry.chen@sinofluoro.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| R&D Scientific Inc. | Canada | |||
|---|---|---|---|---|
 | www.rdscientific.com | |||
 | +1 (226) 600-0236 | |||
 | sales@rdscientific.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Sifluorotm Co,. Ltd. | China | |||
|---|---|---|---|---|
 | www.fluoro.com | |||
 | +86 (0531) 8590-6692 | |||
 | +86 (0531) 8590-6692 | |||
 | inquiry@fluoro.com | |||
 | WeChat: 15288857388 | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2025 | ||||
| P and M Invest Ltd. | Russia | |||
|---|---|---|---|---|
 | www.fluorine.ru | |||
 | +7 (495) 135-6494 | |||
 | +7 (495) 135-6509 | |||
 | sales@fluorine1.ru | |||
| Chemical manufacturer | ||||
| Ricci Chimica | Italy | |||
|---|---|---|---|---|
 | www.riccichimica.com | |||
 | +39 (75) 692-9241 | |||
 | +39 (75) 592-6595 | |||
 | info@riccichimica.com | |||
| Chemical manufacturer | ||||
| MP Biomedicals LLC. | USA | |||
|---|---|---|---|---|
 | www.mpbio.com | |||
 | +1 (440) 337-1200 | |||
 | +1 (440) 337-1180 | |||
 | inside@mpbio.com | |||
| Chemical manufacturer | ||||
| Melford Laboratories Limited | UK | |||
|---|---|---|---|---|
 | www.melford.co.uk | |||
 | +44 (1449) 741-178 | |||
 | +44 (1449) 741-217 | |||
 | pfranklin@melford.co.uk sales@melford.co.uk | |||
| Chemical manufacturer since 1985 | ||||
| Spectrum Chemical Mfg. Corp. | USA | |||
|---|---|---|---|---|
 | www.spectrumchemical.com | |||
 | +1 (310) 516-8000 | |||
 | +1 (310) 516-7512 | |||
 | sales@spectrumchemical.com | |||
| Chemical manufacturer | ||||
| Apollo Scientific Ltd. | UK | |||
|---|---|---|---|---|
 | www.apolloscientific.co.uk | |||
 | +44 (161) 406-0505 | |||
 | +44 (161) 406-0506 | |||
 | sales@apolloscientific.co.uk | |||
| Chemical manufacturer | ||||
| Strem Chemicals, Inc. | USA | |||
|---|---|---|---|---|
 | www.strem.com | |||
 | +1 (978) 499-1600 | |||
 | +1 (978) 465-3104 | |||
 | info@strem.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Halogenated aliphatic hydrocarbon |
|---|---|
| Name | 1,1,1,5,5,5-Hexafluoropentane-2,4-dione |
| Synonyms | Hexafluoroacetylacetone |
| Molecular Structure | 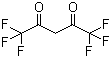 |
| Molecular Formula | C5H2F6O2 |
| Molecular Weight | 208.06 |
| CAS Registry Number | 1522-22-1 |
| EC Number | 216-191-0 |
| SMILES | C(C(=O)C(F)(F)F)C(=O)C(F)(F)F |
| Density | 1.5±0.1 g/cm3, Calc.*, 1.486 g/mL (Expl.) |
|---|---|
| Index of Refraction | 1.305, Calc.*, 1.332 (Expl.) |
| Boiling Point | 70.0 °C (760 mmHg), Calc.*, 70-71 °C (Expl.) |
| Flash Point | 30.8±20.1 °C, Calc.*, 32 °C (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |     GHS02;GHS05;GHS06;GHS07 Danger Details GHS02;GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H226-H301-H302-H312-H314-H318-H332-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P260-P261-P264-P264+P265-P270-P271-P280-P301+P316-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P303+P361+P353-P304+P340-P305+P354+P338-P316-P317-P319-P321-P330-P362+P364-P363-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2920 | ||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
1,1,1,5,5,5-Hexafluoropentane-2,4-dione (C5H2F6O2) is a fluorinated β-diketone widely utilized in coordination chemistry, materials science, and various chemical synthesis applications. The compound is structurally characterized by the presence of two carbonyl groups at the 2- and 4-positions, along with hexafluoroalkyl (-CF3) substituents at the terminal positions. This unique structural motif influences its physicochemical properties, including high volatility, acidity, and strong metal-chelating ability. The synthesis of 1,1,1,5,5,5-hexafluoropentane-2,4-dione follows established methodologies, typically involving the condensation of hexafluoroacetone with acetic acid derivatives or equivalent precursors. The electron-withdrawing nature of the trifluoromethyl groups enhances the acidity of the enol form, leading to its high reactivity in coordination and organic reactions. A major application of this compound lies in metal complexation. As a strong ligand, it forms stable chelates with various metal ions, including transition metals and lanthanides. These complexes are widely used in catalysis, luminescent materials, and as precursors for vapor-phase deposition techniques such as metal-organic chemical vapor deposition (MOCVD). The high volatility and thermal stability of these fluorinated chelates make them suitable for thin-film deposition and advanced material processing. In organic synthesis, 1,1,1,5,5,5-hexafluoropentane-2,4-dione serves as an efficient fluorinated building block. Its ability to undergo nucleophilic substitution, enolate formation, and metal-catalyzed coupling reactions enables the introduction of fluorinated diketone structures into pharmaceuticals, agrochemicals, and specialty polymers. The presence of fluorine atoms influences the lipophilicity and metabolic stability of derived compounds, properties that are often desirable in medicinal chemistry. Due to its volatility and potential reactivity, proper handling and storage are necessary to maintain stability and minimize exposure risks. The compound should be kept in tightly sealed containers under controlled temperature conditions. When working with it in laboratory or industrial settings, appropriate safety measures, including protective equipment and adequate ventilation, are required. The continued interest in fluorinated β-diketones, particularly in the context of advanced materials and metal complexation, ensures the ongoing relevance of 1,1,1,5,5,5-hexafluoropentane-2,4-dione. Its well-established role in coordination chemistry, catalysis, and material science highlights its importance in modern chemical applications. References 2024. Synthesis and Structure of Ag(I) Complexes with Tris(3,5-Dimethylpyrazol-1-yl)Methane and β-Diketones. Journal of Structural Chemistry, 65(11). DOI: 10.1134/s0022476624110180 2024. Direct Effect of Fast Electrons on Hexafluoroacetylacetone. High Energy Chemistry, 58(4). DOI: 10.1134/s0018143924700371 2024. The First Example of Dicubane Nickel(II) Complex in the Series of Unsymmetrically Substituted Diketones. Russian Journal of Coordination Chemistry, 50(7). DOI: 10.1134/s1070328424600499 |
| Market Analysis Reports |