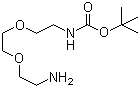
tert-Butyl 2-(2-(2-aminoethoxy)ethoxy)ethylcarbamate is an organic compound belonging to the class of carbamate derivatives. It contains a tert-butyl carbamate (Boc) protecting group, which is commonly used in organic synthesis to protect amine functionalities. The molecular structure consists of an ethoxy-ethoxy-ethyl chain linked to a carbamate functional group, contributing to its solubility and reactivity in various chemical transformations.
The compound is primarily used in synthetic organic chemistry as an intermediate in the preparation of biologically active molecules. The Boc protecting group provides stability to the amine functionality during multi-step synthetic procedures and can be selectively removed under acidic conditions to reveal the free amine for further modifications. This makes it particularly useful in peptide synthesis, pharmaceutical research, and the development of specialized organic compounds.
The discovery and application of Boc-protected amines, including tert-butyl 2-(2-(2-aminoethoxy)ethoxy)ethylcarbamate, are well-documented in the literature. The Boc protection strategy was introduced in the mid-20th century and has since become a widely used method for amine protection due to its compatibility with a broad range of reaction conditions.
In addition to its role as a synthetic intermediate, the compound can be utilized in medicinal chemistry for the modification of drug candidates, particularly in the development of prodrugs or targeted delivery systems where amine functionalities need temporary protection. The presence of the ethoxy-ethoxy-ethyl chain may influence the compound's solubility and reactivity, making it valuable in specific synthetic applications requiring flexible linker systems.
While its applications are mostly confined to laboratory-scale synthesis, the compound’s structural features align with the broader use of Boc-protected amines in chemical research. Proper handling and storage are essential to prevent hydrolysis of the carbamate group under acidic or prolonged exposure to moisture.
References
none
|





















 GHS05;GHS07 Danger Details
GHS05;GHS07 Danger Details