Online Database of Chemicals from Around the World
| Liaoyang Diol Chemicals Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.liaoyangdiol.com | |||
 | +86 (419) 680-5628 +86 13904195924 | |||
 | +86 (419) 413-9769 | |||
 | zhouzhaoxi40@sina.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tyger Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.tygersci.com | |||
 | +1 (609) 434-0144 | |||
 | +1 (609) 434-0143 | |||
 | sales@tygersci.com | |||
| Chemical manufacturer since 1992 | ||||
| chemBlink Standard supplier since 2008 | ||||
| YURUI (Shanghai) Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.riyngroup.com | |||
 | +86 (21) 3319-1321 ex 816 +86 13032131612 | |||
 | +86 (21) 6085-3441 | |||
 | yu@riyngroup.com info@riyngroup.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Yantai Jiacheng Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ytjcchem.com | |||
 | +86 (535) 630-3697 +86 18660586989 | |||
 | +86 (535) 630-3697 | |||
 | sales@ytjcchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Speranza Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.speranzachem.com | |||
 | +86 (755) 3312-2716 | |||
 | +86 (755) 3312-2715 | |||
 | sales@speranzachem.com | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Carbosynth China Ltd. | China | |||
|---|---|---|---|---|
 | www.carbosynth.cn | |||
 | +86 (512) 6260-5585 | |||
 | +86 (512) 6260-5576 | |||
 | sales@carbosynth.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zhejiang Free Trade Zone Violet Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjviolettech.com | |||
 | +86 (580) 811-8992 | |||
 | +86 (580) 811-9487 | |||
 | jenny@zjviolettech.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Aajiang Jiuzhou Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jiuzhou-chem.com | |||
 | +86 13454675544 | |||
 | jamie@jiuzhou-chem.com | |||
 | QQ Chat | |||
 | WeChat: +86 18632988332 | |||
 | WhatsApp:+86 18632988332 | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2023 | ||||
| NNU Chemlab613 | China | |||
|---|---|---|---|---|
 | www.chemlab613.com | |||
 | +86 (25) 5874-1998 | |||
 | +86 (25) 5874-8998 | |||
 | news@chemlab613.com | |||
| Chemical manufacturer since 2003 | ||||
| Atlantic Research Chemicals Ltd. | UK | |||
|---|---|---|---|---|
 | www.atlantic-chemicals.com | |||
 | +44 (8707) 746-454 | |||
 | +44 (8707) 746-455 | |||
 | info@atlantic-chemicals.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Hydrocarbon halide |
|---|---|
| Name | 4-Iodobiphenyl |
| Synonyms | 1-iodo-4-phenylbenzene |
| Molecular Structure | 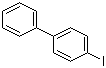 |
| Molecular Formula | C12H9I |
| Molecular Weight | 280.11 |
| CAS Registry Number | 1591-31-7 |
| EC Number | 216-469-1 |
| SMILES | C1=CC=C(C=C1)C2=CC=C(C=C2)I |
| Melting point | 110-114 °C |
|---|---|
| Hazard Symbols |    GHS05;GHS07;GHS09 Danger Details GHS05;GHS07;GHS09 Danger Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H318-H400-H410 Details | ||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P273-P280-P301+P317-P305+P354+P338-P317-P330-P391-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| Transport Information | UN 3077 | ||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
4-Iodobiphenyl is a significant chemical compound in organic chemistry, valued for its role in various chemical reactions and applications. This compound consists of two biphenyl rings connected by an iodine atom at the para position, which imparts unique properties to the molecule. The discovery of 4-iodobiphenyl can be traced back to early studies in organoiodine chemistry, where researchers were exploring the reactivity and applications of iodine-substituted aromatic compounds. Iodine, as a substituent, introduces distinct electronic and steric effects due to its size and electronegativity, which can greatly influence the chemical behavior of the biphenyl system. 4-Iodobiphenyl is primarily used as an intermediate in the synthesis of more complex molecules. Its role as a versatile building block is crucial in the production of pharmaceuticals, agrochemicals, and materials science. One of its prominent applications is in the synthesis of ligands for palladium-catalyzed cross-coupling reactions, such as the Suzuki-Miyaura reaction. In this reaction, 4-iodobiphenyl acts as a key component that couples with other organoboron compounds to form biaryl systems, which are valuable in various chemical and pharmaceutical applications. In addition to its role in cross-coupling reactions, 4-iodobiphenyl is also utilized in the creation of liquid crystals. Liquid crystals are materials that exhibit properties between those of conventional liquids and solid crystals, making them useful in display technologies. The incorporation of 4-iodobiphenyl into liquid crystal formulations can modify the alignment and properties of the liquid crystals, enhancing their performance in devices such as LCD screens. The compound's utility extends to organic synthesis, where it is employed in the preparation of functionalized biphenyl derivatives. These derivatives are important in the development of new materials and chemical products. The reactivity of 4-iodobiphenyl, due to the presence of the iodine atom, allows for various transformations that are not possible with other biphenyl derivatives. In summary, 4-iodobiphenyl is a valuable compound in organic chemistry, recognized for its role in both industrial and research applications. Its ability to participate in cross-coupling reactions and its application in liquid crystal technology highlight its importance in the synthesis of complex molecules and advanced materials. References 1981. Biodehalogenation and Metabolism of [125I]-4-iodobiphenyl. Journal of Pharmaceutical Sciences, 70(5), 521-526. DOI: https://pubmed.ncbi.nlm.nih.gov/7241361 2020. Advances in Carbon-Element Bond Construction under Chan-Lam Cross-Coupling Conditions: A Second Decade. Synthesis, 52(24), 3767-3795. DOI: 10.1055/s-0040-1705971 2022. Copper-mediated etherification via aryl radicals generated from triplet states. Nature Synthesis, 1(5), 393-402. DOI: 10.1038/s44160-022-00061-0 |
| Market Analysis Reports |