Online Database of Chemicals from Around the World
| Luminescence Technology Corp. | Taiwan | |||
|---|---|---|---|---|
 | www.lumtec.com.tw | |||
 | +886 (2) 2697-5600 | |||
 | +886 (2) 2697-5601 | |||
 | sales@lumtec.com.tw | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2024 | ||||
| Classification | Pharmaceutical intermediate >> OLED material intermediate |
|---|---|
| Name | 10-(4-(4,6-Diphenyl-1,3,5-triazin-2-yl)phenyl)-9,9-dimethyl-9,10-dihydroacridine |
| Synonyms | DMAC-TRZ |
| Molecular Structure | 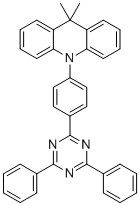 |
| Molecular Formula | C36H28N4 |
| Molecular Weight | 516.63 |
| CAS Registry Number | 1628752-98-6 |
| EC Number | 891-272-7 |
| SMILES | CC1(C2=CC=CC=C2N(C3=CC=CC=C31)C4=CC=C(C=C4)C5=NC(=NC(=N5)C6=CC=CC=C6)C7=CC=CC=C7)C |
| Solubility | soluble (toluene) |
|---|---|
| Density | 1.2±0.1 g/cm3, Calc.* |
| Index of Refraction | 1.652, Calc.* |
| Boiling Point | 716.3±70.0 °C (760 mmHg), Calc.* |
| Flash Point | 387.0±35.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
10-(4-(4,6-Diphenyl-1,3,5-triazin-2-yl)phenyl)-9,9-dimethyl-9,10-dihydroacridine is a unique chemical compound that belongs to the class of organic molecules known for their complex structure and potential applications in various fields, particularly in materials science and organic electronics. This molecule consists of a modified acridine core substituted with both a diphenyltriazinyl and a dimethyl group, giving it distinct electronic properties and making it suitable for several advanced applications. The discovery of 10-(4-(4,6-Diphenyl-1,3,5-triazin-2-yl)phenyl)-9,9-dimethyl-9,10-dihydroacridine is a result of efforts to design and synthesize molecules that can exhibit high stability and efficient charge transport properties. The incorporation of the 4,6-diphenyl-1,3,5-triazine unit into the structure serves to enhance the molecule's electronic characteristics by improving electron delocalization and creating conjugation pathways that are beneficial for charge transfer. Additionally, the dimethyl group at the acridine position increases the molecule's stability and solubility, making it easier to incorporate into various devices. One of the most significant applications of this compound lies in the field of organic electronics, particularly in organic light-emitting diodes (OLEDs) and organic photovoltaic (OPV) devices. The triazine unit acts as an electron-accepting group, while the acridine core provides a platform for efficient electron transport. This combination of donor-acceptor interactions enables the molecule to function effectively in electronic devices, where charge separation and transfer are critical for performance. The diphenyltriazinyl group also contributes to the molecule's stability under electrical and thermal stress, which is important for the longevity and efficiency of OLEDs and OPVs. In addition to organic electronics, this compound has shown potential in the development of molecular materials for use in molecular sensors and optoelectronic devices. The tunability of the electronic properties, as well as the ability to modify the molecule's structure further, makes it a promising candidate for a wide range of applications. Research has demonstrated that the interaction between the triazine and acridine units can lead to favorable optical and electronic properties, which can be fine-tuned by altering the substituent groups or the core structure itself. The synthesis of 10-(4-(4,6-Diphenyl-1,3,5-triazin-2-yl)phenyl)-9,9-dimethyl-9,10-dihydroacridine involves the careful incorporation of the triazine and dimethyl acridine units through well-established organic synthetic methodologies, including nucleophilic substitution and condensation reactions. These synthetic routes allow for the precise control of the electronic structure and functional groups, which are critical for achieving the desired properties of the final compound. In summary, 10-(4-(4,6-Diphenyl-1,3,5-triazin-2-yl)phenyl)-9,9-dimethyl-9,10-dihydroacridine is a promising organic compound with unique electronic properties that make it suitable for applications in organic electronics, molecular sensors, and optoelectronic devices. Its stability, electronic tunability, and efficient charge transport capabilities position it as a key candidate for the development of advanced materials in next-generation electronic technologies. References 2024. Charge trapping for controllable persistent luminescence in organics. Nature Photonics, 18(4), 338-346. DOI: 10.1038/s41566-024-01396-0 2021. Thermally activated delayed fluorescence (TADF) organic molecules for efficient X-ray scintillation and imaging. Nature Materials, 21(2), 210-216. DOI: 10.1038/s41563-021-01132-x |
| Market Analysis Reports |