Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| TH Chemica Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.thchemica.com | |||
 | +86 (573) 8283-5115 +86 13998887555 | |||
 | +86 (573) 8396-5652 | |||
 | thchemica@126.com info@thchemica.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2009 | ||||
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Relybo Pharmachemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.relybopharma.com | |||
 | +86 (25) 8771-6566 | |||
 | susanchen@relybopharma.com | |||
| Chemical distributor since 2011 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Ensky Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.ensky-chemical.com | |||
 | +86 (23) 8681-5286 | |||
 | +86 (23) 8681-5116 | |||
 | sales@ensky-chemical.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Kessie Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kessiechem.com | |||
 | +86 (519) 8698-7916 +86 15851999766 | |||
 | +86 (519) 8690-8212 | |||
 | info@kessiechem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Hangzhou Zentra Bio-Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zentrachem.com | |||
 | 18321999949 | |||
 | steven@zentrachem.com | |||
 | QQ Chat | |||
 | WeChat: Hangzhou Zentra Bio-Chemical Co., Ltd. | |||
| Chemical distributor since 2025 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Wako Pure Chemical Industries, Ltd. | Japan | |||
|---|---|---|---|---|
 | www.wako-chem.co.jp | |||
 | +81 (6) 6203-3741 | |||
 | +81 (6) 6201-5964 | |||
 | wkhk.info@fujifilm.com | |||
| Chemical manufacturer since 1922 | ||||
| Biofine International Inc. | USA | |||
|---|---|---|---|---|
 | www.biofineintl.com | |||
 | +1 (604) 438-8181 | |||
 | +1 (604) 438-8378 | |||
 | info@biofineintl.com | |||
| Chemical manufacturer | ||||
| W & J PharmaChem, Inc. | USA | |||
|---|---|---|---|---|
 | www.wjpharmachem.com | |||
 | +1 (301) 880-0624 | |||
 | +1 (301) 434-9366 | |||
 | info@wjpharmachem.com | |||
| Chemical manufacturer | ||||
| Ryan Scientific, Inc. | USA | |||
|---|---|---|---|---|
 | www.ryansci.com | |||
 | +1 (843)-884-4911 | |||
 | +1 (843) 884-5568 | |||
 | sales@ryansci.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Heterocyclic compound >> Indoles |
|---|---|
| Name | 2-(Ethoxycarbonyl)-5-bromo-indole |
| Synonyms | 5-Bromoindole-2-carboxylic acid ethyl ester |
| Molecular Structure | 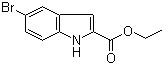 |
| Molecular Formula | C11H10BrNO2 |
| Molecular Weight | 268.11 |
| CAS Registry Number | 16732-70-0 |
| EC Number | 680-051-9 |
| SMILES | CCOC(=O)C1=CC2=C(N1)C=CC(=C2)Br |
| Density | 1.6±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 163-167 °C (Expl.) |
| Index of Refraction | 1.646, Calc.* |
| Boiling Point | 394.7±22.0 °C (760 mmHg), Calc.* |
| Flash Point | 192.5±22.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
2-(Ethoxycarbonyl)-5-bromo-indole is a valuable compound in organic chemistry, characterized by an indole backbone substituted with an ethoxycarbonyl group at the 2-position and a bromine atom at the 5-position. This dual substitution imparts unique reactivity to the molecule, making it a useful intermediate in pharmaceutical and material science applications. The discovery of 2-(ethoxycarbonyl)-5-bromo-indole was driven by the need for functionalized indole derivatives that could be further modified for specialized applications. Indole derivatives have long been recognized for their importance in natural products, pharmaceuticals, and agrochemicals. The introduction of an ethoxycarbonyl group enhances the molecule's versatility in coupling and condensation reactions, while the bromine atom facilitates halogen-specific transformations, such as Suzuki-Miyaura or Stille coupling reactions. In the pharmaceutical field, 2-(ethoxycarbonyl)-5-bromo-indole serves as a precursor in the synthesis of biologically active molecules. Its indole core, a structural motif found in many natural and synthetic drugs, allows it to participate in reactions leading to anti-cancer, anti-inflammatory, and anti-viral agents. The ethoxycarbonyl group can be selectively transformed, enabling the creation of tailored derivatives with improved bioactivity and pharmacokinetic properties. For instance, this compound has been explored in the development of kinase inhibitors and serotonin receptor modulators. In materials science, 2-(ethoxycarbonyl)-5-bromo-indole is employed in the design of advanced organic semiconductors and fluorescent materials. The indole ring system, known for its electronic properties, can be incorporated into π-conjugated systems. By utilizing the bromine atom for cross-coupling reactions, researchers have synthesized indole-based polymers and small molecules that exhibit excellent optical and electronic behavior. These materials find applications in organic light-emitting diodes (OLEDs), solar cells, and field-effect transistors. Synthetic strategies for 2-(ethoxycarbonyl)-5-bromo-indole typically involve the bromination of ethyl indole-2-carboxylate under controlled conditions to achieve selective functionalization at the 5-position. Advances in synthetic methodologies aim to improve reaction efficiency, reduce the use of hazardous reagents, and minimize by-products. These efforts align with the growing emphasis on green chemistry and sustainable industrial practices. The handling of 2-(ethoxycarbonyl)-5-bromo-indole requires standard precautions due to the reactivity of its brominated and ester functionalities. Safe storage and disposal practices are essential to prevent environmental contamination and ensure laboratory safety. In summary, 2-(ethoxycarbonyl)-5-bromo-indole is a versatile compound with significant contributions to pharmaceutical development and materials innovation. Its structural attributes and reactivity make it a cornerstone for designing new compounds and materials with tailored properties. References 2016. Design, synthesis, and antimicrobial activity of new 5-substituted indole-2-carboxamide derivatives. Research on Chemical Intermediates. DOI: 10.1007/s11164-016-2696-3 |
| Market Analysis Reports |