Online Database of Chemicals from Around the World
| Nivon Specialties | India | |||
|---|---|---|---|---|
 | www.navoneindia.com | |||
 | +91 9323789882 | |||
 | nivonpharma@yahoo.com | |||
| Chemical manufacturer since 1999 | ||||
| chemBlink Standard supplier since 2025 | ||||
| Classification | Chemical reagent >> Organic reagent >> Amide |
|---|---|
| Name | Rebemide |
| Synonyms | N,N-diethylbenzamide |
| Molecular Structure | 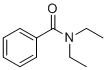 |
| Molecular Formula | C11H15NO |
| Molecular Weight | 177.24 |
| CAS Registry Number | 1696-17-9 |
| EC Number | 216-912-9 |
| SMILES | CCN(CC)C(=O)C1=CC=CC=C1 |
| Density | 1.0±0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 28 - 32 °C (Expl.) |
| Boiling point | 288.7±9.0 °C 760 mmHg (Calc.)*, 299 - 304.4 °C (Expl.) |
| Flash point | 125.5±9.9 °C (Calc.)* |
| Index of refraction | 1.518 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H318-H335 Details | ||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P354+P338-P317-P319-P321-P330-P332+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
Rebemide is a cytoprotective and mucosal-protective agent that has been investigated and used for the treatment of gastric mucosal injuries, particularly those caused by non-steroidal anti-inflammatory drugs (NSAIDs), stress, or alcohol. Its chemical name is 4-(4-chlorobenzoyl)-N,N-dimethyl-5-oxo-1,2,3,5-tetrahydro-1H-pyrrolo[1,2-a]imidazole-7-carboxamide, and its molecular formula is C16H16ClN3O3. Rebemide was first developed in Japan in the 1990s as part of efforts to find therapeutic alternatives that protect the gastrointestinal tract without affecting gastric acid secretion, as is common with proton pump inhibitors and H2 receptor antagonists. Unlike acid suppressants, rebemide does not significantly alter gastric pH but instead enhances the protective mechanisms of the gastric mucosa. The compound exerts its effects through several mechanisms. It stimulates the secretion of gastric mucus and increases the synthesis of prostaglandins in the gastric lining, both of which are essential for maintaining mucosal integrity. Additionally, it has been shown to improve mucosal blood flow and inhibit neutrophil activation, thereby reducing inflammation and oxidative damage in the gastric tissues. These properties make it useful in preventing mucosal injury in patients undergoing NSAID therapy or those with stress-related mucosal disease. Rebemide is administered orally and is absorbed through the gastrointestinal tract. Pharmacokinetic studies in humans have shown that the drug reaches peak plasma concentration within a few hours of administration and is eliminated primarily via renal excretion. Its safety profile is considered favorable, with low incidence of adverse effects reported in clinical studies. Common side effects, when they occur, are mild and may include gastrointestinal discomfort or headache. In clinical trials, rebemide has demonstrated efficacy in healing gastric erosions and ulcers and in reducing the recurrence of lesions during continued NSAID use. It is also of interest in the management of gastritis and other forms of non-ulcer dyspepsia due to its protective and anti-inflammatory effects on the gastric mucosa. Although primarily used in certain East Asian countries, particularly Japan, rebemide has not been widely adopted or approved in many Western nations. Nonetheless, its development represents a notable effort toward cytoprotective strategies in gastroenterology that do not rely solely on acid suppression. In summary, rebemide is a gastric mucosal-protective agent that enhances the body's natural defense mechanisms in the stomach lining. It is used to prevent and treat gastric injuries, particularly those associated with NSAIDs, and represents a therapeutic alternative in cases where acid suppression is insufficient or inappropriate. References 2018. Electrophilic Activation of Carboxylic Anhydrides for Nucleophilic Acylation Reactions. Synthesis, 50(16). DOI: 10.1055/s-0037-1609564 2017. Formation of amides: one-pot condensation of carboxylic acids and amines mediated by TiCl4. Chemistry Central Journal, 11(1). DOI: 10.1186/s13065-017-0318-9 2016. Metal-Free Amidation of Acids with Formamides and T3P®. Synthesis, 48(12). DOI: 10.1055/s-0035-1561427 |
| Market Analysis Reports |