Online Database of Chemicals from Around the World
| BrightGene Bio-medical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bright-gene.com | |||
 | +86 (512) 6255-1801 6255-1767 +86 13812696362 | |||
 | +86 (512) 6255-1799 | |||
 | kevinwan@bright-gene.com kevinwan0203@gmail.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2009 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shenzhen JYMed Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jymedtech.com | |||
 | +86 (755) 2661-2112 | |||
 | +86 (755) 8635-0488 | |||
 | jymed@jymedtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Shanghai Zehan Biopharma Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zehanbiopharma.com | |||
 | +86 (21) 6135-0663 +86 13052117465 | |||
 | +86 (21) 6135-0662 | |||
 | sales@zehanbiopharma.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | API >> Antibiotics |
|---|---|
| Name | Oritavancin |
| Synonyms | (4''R)-22-O-(3-Amino-2,3,6-trideoxy-3-C-methyl-alpha-L-arabino-hexopyranosyl)-N3''-[(4'-chloro[1,1'-biphenyl]-4-yl)methyl]vancomycin; LY 333328; Orbactiv |
| Molecular Structure | 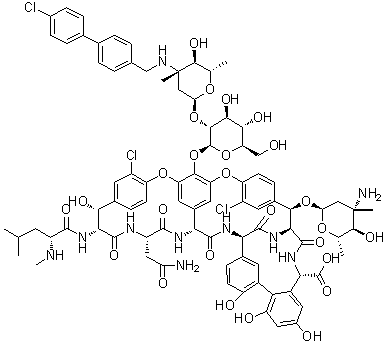 |
| Molecular Formula | C86H97Cl3N10O26 |
| Molecular Weight | 1793.10 |
| CAS Registry Number | 171099-57-3 |
| SMILES | C[C@H]1[C@@H]([C@@](C[C@@H](O1)O[C@H]2[C@H]3C(=O)N[C@@H](C4=C(C(=CC(=C4)O)O)C5=C(C=CC(=C5)[C@H](C(=O)N3)NC(=O)[C@H]6C7=CC(=C(C(=C7)OC8=C(C=C2C=C8)Cl)O[C@H]9[C@@H]([C@H]([C@@H]([C@H](O9)CO)O)O)O[C@H]1C[C@]([C@H]([C@@H](O1)C)O)(C)NCC1=CC=C(C=C1)C1=CC=C(C=C1)Cl)OC1=C(C=C(C=C1)[C@H]([C@H](C(=O)N[C@H](C(=O)N6)CC(=O)N)NC(=O)[C@@H](CC(C)C)NC)O)Cl)O)C(=O)O)(C)N)O |
| Solubility | Insoluble (2.0E-5 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.59±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.724, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
|
Oritavancin is a semisynthetic lipoglycopeptide antibiotic developed for the treatment of serious bacterial infections, particularly those caused by Gram-positive pathogens. Its discovery was driven by the need for more effective agents against multidrug-resistant bacteria, leading to the modification of natural glycopeptides to enhance their activity. Oritavancin was derived from chloroeremomycin, a fermentation product of *Actinobacteria*, through structural modifications that improved its potency and pharmacokinetic properties. It was approved by the United States Food and Drug Administration in 2014 for the treatment of acute bacterial skin and skin structure infections (ABSSSI). The mechanism of action of oritavancin involves multiple pathways, making it highly effective against resistant bacteria, including methicillin-resistant *Staphylococcus aureus* (MRSA) and vancomycin-resistant *Enterococcus* (VRE). It inhibits bacterial cell wall synthesis by binding to the D-Ala-D-Ala terminal of peptidoglycan precursors, disrupting transpeptidation and transglycosylation. Additionally, it disrupts membrane integrity, leading to rapid bacterial cell death. These combined actions contribute to its strong bactericidal properties and reduce the likelihood of resistance development. The primary clinical application of oritavancin is in the treatment of skin and soft tissue infections caused by Gram-positive organisms. Its long half-life allows for a single-dose intravenous administration, providing a significant advantage over traditional antibiotics that require multiple doses over several days. This dosing regimen improves patient compliance and reduces hospitalization costs. Oritavancin has also been studied for its potential use in bloodstream infections, osteomyelitis, and prosthetic joint infections, where its potent activity against resistant pathogens is particularly valuable. Research continues to explore the broader applications of oritavancin, including its use in combination therapies and its effectiveness against emerging antibiotic-resistant bacteria. Its unique mechanism of action and extended duration of activity make it a promising candidate for future developments in antimicrobial therapy. Despite its effectiveness, the high cost and limited spectrum against Gram-negative bacteria necessitate careful selection of patients to maximize clinical benefits. References 1996. Reductive Alkylation of Glycopeptide Antibiotics: Synthesis and Antibacterial Activity. The Journal of Antibiotics, 49(6). DOI: 10.7164/antibiotics.49.575 2023. TTD: Therapeutic Target Database describing target druggability information. Nucleic Acids Research, 52(D1). DOI: 10.1093/nar/gkad751 2021. Strategy for Producing the High-Quality Glycopeptide Antibiotic A82846B in Amycolatopsis orientalis Based on the CRISPR-Cas12a System. ACS Synthetic Biology, 10(10). DOI: 10.1021/acssynbio.1c00317 |
| Market Analysis Reports |