Online Database of Chemicals from Around the World
| Gou Hebei Chemicals | India | |||
|---|---|---|---|---|
 | gouhebeichems.store | |||
 | +86 15874272473 | |||
 | info@gouhebeichems.store | |||
| Chemical distributor since 2009 | ||||
| chemBlink Standard supplier since 2023 | ||||
| Classification | Natural product >> Terpenes |
|---|---|
| Name | (+/-)-5-Fluoro-adb |
| Synonyms | methyl 2-[[1-(5-fluoropentyl)indazole-3-carbonyl]amino]-3,3-dimethylbutanoate |
| Molecular Structure | 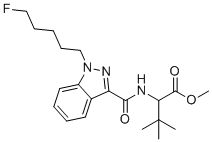 |
| Molecular Formula | C20H28FN3O3 |
| Molecular Weight | 377.45 |
| CAS Registry Number | 1715016-75-3 |
| EC Number | 822-744-2 |
| SMILES | CC(C)(C)C(C(=O)OC)NC(=O)C1=NN(C2=CC=CC=C21)CCCCCF |
| Density | 1.2±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.546, Calc.* |
| Boiling Point | 551.9±40.0 °C (760 mmHg), Calc.* |
| Flash Point | 287.6±27.3 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H319--H335-H336 Details | ||||||||
| Safety Statements | P261-P264+P265-P271-P280-P304+P340-P305+P351+P338-P319-P337+P317-P403+P233-P405-P501 Details | ||||||||
| Hazard Classification | |||||||||
| |||||||||
|
(+/-)-5-Fluoro-ADB, also known as 5-Fluoro-ADB-PINACA, is a synthetic cannabinoid that has attracted attention for its potent psychoactive effects and growing popularity in the recreational drug market. As a fluorinated analog of ADB-PINACA, the compound interacts with cannabinoid receptors to produce effects similar to delta-9-tetrahydrocannabinol (THC), the active ingredient in marijuana. The development of (+/-)-5-Fluoro-ADB stems from the quest for novel synthetic cannabinoids that could circumvent legal restrictions and replicate or enhance the effects of natural cannabinoids. Researchers initially synthesized fluorinated analogs of known synthetic cannabinoids to study their pharmacological properties and assess their potential as therapeutic or recreational drugs. Fluorine substitutions were introduced in (+/-)-5-Fluoro-ADB to enhance its binding affinity and metabolic stability, resulting in a potent compound with significant psychoactive properties. The indazole core is a central structure common to many synthetic cannabinoids and is essential for binding to cannabinoid receptors. Fluorine substitutions on the pentyl chain increase binding affinity and metabolic resistance. The amide group enhances stability and potency, contributing to the pharmacological activity of the compound. (+/-)-5-Fluoro-ADB is used to study the interaction of synthetic cannabinoids with cannabinoid receptors (CB1 and CB2). These studies help to elucidate the binding affinity, efficacy, and potency of synthetic cannabinoids, providing insight into their pharmacological actions and potential therapeutic applications. The compound is valuable in SAR studies to understand the effects of fluorine substitution on the activity of synthetic cannabinoids. This knowledge can aid in the design of new compounds with desirable pharmacological characteristics for potential medical or recreational use. Due to its presence in the illicit drug market, (+/-)-5-Fluoro-ADB is frequently tested for drug testing and toxicological analysis in forensic laboratories. Methods such as gas chromatography-mass spectrometry (GC-MS) and liquid chromatography-tandem mass spectrometry (LC-MS/MS) are used to identify and quantify the compound in biological samples and seized drugs. Researchers study the toxicological effects of (+/-)-5-Fluoro-ADB to assess its safety and potential health risks. These studies are critical to understanding the adverse effects associated with synthetic cannabinoid use and to informing public health policy and regulatory decisions. (+/-)-5-Fluoro-ADB is often marketed as a designer drug or synthetic alternative to cannabis. Its powerful psychoactive effects are similar to those of natural cannabinoids, making it popular among recreational users. However, its use is associated with significant health risks, including severe intoxication, psychosis, and even death. The emergence of (+/-)-5-Fluoro-ADB highlights the challenges regulators face in controlling synthetic cannabinoids. Its legal status varies by region, and its rapid proliferation highlights the need for a comprehensive and adaptive regulatory framework to address the changing synthetic drug landscape. References 2016. Pharmacology of Valinate and tert-Leucinate Synthetic Cannabinoids 5F-AMBICA, 5F-AMB, 5F-ADB, AMB-FUBINACA, MDMB-FUBINACA, MDMB-CHMICA, and Their Analogues. ACS Chemical Neuroscience, 7(9). DOI: 10.1021/acschemneuro.6b00137 2023. Death after smoking of fentanyl, 5F-ADB, 5F-MDMB-P7AICA and other synthetic cannabinoids with a bucket bong. Forensic Toxicology, 41(2). DOI: 10.1007/s11419-023-00666-w 2018. Metabolic fate of the new synthetic cannabinoid 7'N‐5F‐ADB in rat, human, and pooled human S9 studied by means of hyphenated high‐resolution mass spectrometry. Drug Testing and Analysis, 11(2). DOI: 10.1002/dta.2493 |
| Market Analysis Reports |