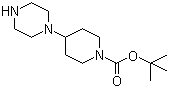
4-Piperazin-1-ylpiperidine-1-carboxylic acid tert-butyl ester is a synthetic intermediate that has played a significant role in the development of medicinal chemistry, particularly in the design of bioactive molecules targeting central nervous system receptors. This compound features a structural combination of a piperidine and a piperazine ring, connected through a nitrogen atom, with a tert-butyl ester group protecting the carboxyl functionality. The presence of two nitrogen-containing rings provides multiple sites for functionalization, making it a highly versatile scaffold for drug discovery efforts.
The compound was initially synthesized during broader studies focused on modifying polycyclic and heterocyclic frameworks to enhance pharmacological profiles. The incorporation of the piperazine moiety is particularly important because piperazine is a privileged structure in medicinal chemistry, known for its presence in numerous drugs with diverse activities, including antipsychotic, antidepressant, and antihistamine effects. Similarly, the piperidine ring contributes to receptor binding affinity and metabolic stability. The tert-butyl ester group protects the carboxyl group during synthetic transformations, allowing selective reactions elsewhere on the molecule before final deprotection steps.
In pharmaceutical research, 4-piperazin-1-ylpiperidine-1-carboxylic acid tert-butyl ester has been employed as a critical intermediate for synthesizing ligands targeting neurotransmitter systems, particularly dopaminergic and serotonergic receptors. These receptors are implicated in the pathophysiology of various neuropsychiatric disorders such as schizophrenia, depression, and anxiety. By modifying this core structure with different substituents on the piperazine or piperidine rings, researchers have been able to modulate the binding properties and selectivity of the resulting compounds, leading to the identification of potential therapeutic candidates.
Beyond central nervous system applications, derivatives of this compound have also been explored for their potential in oncology. Small molecule inhibitors designed to interfere with protein-protein interactions often employ nitrogen-rich heterocycles to optimize binding affinity and specificity. The dual nitrogen atoms from the piperidine and piperazine rings offer the possibility of engaging in hydrogen bonding and electrostatic interactions with biological targets, thereby enhancing the pharmacological profile of candidate molecules.
The application of 4-piperazin-1-ylpiperidine-1-carboxylic acid tert-butyl ester is not limited to direct therapeutic agents. It has also been used in the synthesis of chemical probes for biological studies, helping researchers elucidate receptor functions and signaling pathways. The modularity of the molecule allows for the attachment of fluorescent tags, affinity labels, or other reporter groups without significantly disrupting the biological properties of the parent scaffold. This adaptability has facilitated its use in both high-throughput screening and detailed mechanistic investigations.
In practical synthetic chemistry, this compound is prized for its chemical stability and compatibility with a wide range of reaction conditions. The tert-butyl ester group can be readily removed under acidic conditions to yield the corresponding free acid, which is often a critical step in the final stages of drug synthesis. Additionally, the secondary amine functionalities in the piperazine and piperidine rings provide convenient sites for alkylation, acylation, or sulfonylation reactions, enabling the generation of a wide array of derivatives tailored for specific biological activities.
The discovery and subsequent application of 4-piperazin-1-ylpiperidine-1-carboxylic acid tert-butyl ester illustrate the importance of designing versatile chemical intermediates that can be readily adapted to diverse synthetic needs. Its development has significantly contributed to the advancement of medicinal chemistry programs, especially in the fields of neuroscience and cancer biology. Today, it continues to be widely used as a starting material in the exploration of new therapeutic agents and molecular probes across various domains of biomedical research.
|












 GHS07 Warning Details
GHS07 Warning Details