Online Database of Chemicals from Around the World
| Shanghai Rui Yun Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.rychemical.com.cn | |||
 | +86 (21) 6726-7633 | |||
 | +86 (21) 6726-7655 | |||
 | sales@rychemical.com.cn | |||
 | WeChat: 13917251563 | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Premium supplier since 2009 | ||||
| Classification | Chemical reagent >> Organic reagent >> Olefins (cyclic and non-cyclic) |
|---|---|
| Name | 4-Bromo-3,3,4,4-tetrafluorobut-1-ene |
| Synonyms | 4-Bromo-3,3,4,4-tetrafluoro-1-butene |
| Molecular Structure | 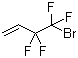 |
| Molecular Formula | C4H3BrF4 |
| Molecular Weight | 206.96 |
| CAS Registry Number | 18599-22-9 |
| EC Number | 242-440-8 |
| SMILES | C=CC(C(F)(F)Br)(F)F |
| Density | 1.659 |
|---|---|
| Boiling point | 81 °C |
| Refractive index | 1.3773 |
| Flash point | -2 °C |
| Hazard Symbols |   GHS02;GHS07 Danger Details GHS02;GHS07 Danger Details | ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H225-H315-H319-H335-H336-H412 Details | ||||||||||||||||||||||||||||
| Safety Statements | P210-P233-P240-P241-P242-P243-P261-P264-P264+P265-P271-P273-P280-P302+P352-P303+P361+P353-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P370+P378-P403+P233-P403+P235-P405-P501 Details | ||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||
|
4-Bromo-3,3,4,4-tetrafluorobut-1-ene is an organofluorine compound characterized by its four fluorine atoms and a bromine atom attached to a butene backbone. This compound is notable for its dual reactivity, combining the high electronegativity and stability of fluorine atoms with the reactivity of bromine. The presence of the fluorine atoms in this structure contributes to the molecule's enhanced stability and resistance to chemical degradation, while the bromine allows for selective reactions, particularly in substitution and addition processes. The discovery of 4-bromo-3,3,4,4-tetrafluorobut-1-ene can be traced to advancements in organofluorine chemistry, where researchers sought to design molecules with specific reactivity patterns that could be harnessed in both synthetic chemistry and material applications. The introduction of fluorine atoms into hydrocarbon chains is known to dramatically influence physical and chemical properties, such as reducing polarizability, increasing lipophobicity, and improving thermal stability. These properties make fluorinated compounds, like this one, attractive intermediates in various fields. One of the primary applications of 4-bromo-3,3,4,4-tetrafluorobut-1-ene is in the synthesis of fluorinated polymers and other materials that require high chemical resistance. Its structure makes it a useful monomer or intermediate in the production of fluoropolymers, which are used in coatings, non-stick surfaces, and specialized industrial materials that demand resistance to heat, chemicals, and degradation. The presence of both fluorine and bromine also makes this compound valuable for further derivatization, particularly in pharmaceutical and agrochemical synthesis where fluorinated groups enhance the biological activity of active molecules. In addition to material science, 4-bromo-3,3,4,4-tetrafluorobut-1-ene is also used in organic synthesis. The bromine atom serves as a reactive site for nucleophilic substitution reactions, enabling the introduction of different functional groups into the fluorinated carbon chain. This versatility makes it an important building block for the production of more complex fluorinated organic compounds, which have applications in medicinal chemistry, especially in the development of drugs that benefit from the stability and metabolic properties imparted by fluorine atoms. The combination of fluorine's unique properties with the reactivity of bromine positions 4-bromo-3,3,4,4-tetrafluorobut-1-ene as an essential component in the expanding field of fluorine chemistry, with ongoing research and industrial applications driving its continued relevance. References 2021. Diversity-Oriented Synthesis of Highly Functionalized Alicycles across Dipolar Cycloaddition/Metathesis Reaction. Synlett. DOI: 10.1055/s-0040-1706041 2020. Synthesis and Application of Tetrafluoroethylene (CF2CF2)-Containing Acetylene Derivatives. Synthesis. DOI: 10.1055/s-0039-1691744 2010. Novel Introduction of a Tetrafluoroethylene (-CF2CF2-) Unit into Organic Molecules. Synthesis. DOI: 10.1055/s-0030-1258330 |
| Market Analysis Reports |