Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Isoquinoline compound |
|---|---|
| Name | (S)-4-(3-bromophenyl)-6,8-dichloro-2-methyl-1,2,3,4-tetrahydroisoquinoline |
| Molecular Structure | 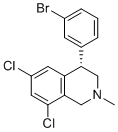 |
| Molecular Formula | C16H14BrCl2N |
| Molecular Weight | 371.10 |
| CAS Registry Number | 1870821-29-6 |
| SMILES | CN1C[C@H](C2=C(C1)C(=CC(=C2)Cl)Cl)C3=CC(=CC=C3)Br |
| Density | 1.5±0.1 g/cm3, Calc.* |
|---|---|
| Index of Refraction | 1.622, Calc.* |
| Boiling Point | 412.8±45.0 °C (760 mmHg), Calc.* |
| Flash Point | 203.4±28.7 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302-H315-H319-H335 Details |
| Safety Statements | P261-P280-P301+P312-P302+P352-P305+P351+P338 Details |
| SDS | Available |
|
(S)-4-(3-Bromophenyl)-6,8-dichloro-2-methyl-1,2,3,4-tetrahydroisoquinoline is an organic compound with the molecular formula C₁₆H₁₄BrCl₂N. It belongs to the class of tetrahydroisoquinolines, characterized by a saturated isoquinoline core structure. The compound features bromine and chlorine substituents on the phenyl and isoquinoline rings, respectively, along with a methyl group. citeturn0search0 The synthesis of this compound typically involves multi-step organic reactions. One common method includes the use of brominated and chlorinated precursors, undergoing a series of reactions such as halogenation, cyclization, and methylation. These reactions often require specific catalysts, solvents, and controlled temperatures to achieve the desired stereochemistry and yield. citeturn0search1 While detailed information on the discovery and applications of (S)-4-(3-Bromophenyl)-6,8-dichloro-2-methyl-1,2,3,4-tetrahydroisoquinoline is limited in publicly available literature, compounds within the tetrahydroisoquinoline class have been studied for various pharmacological activities. For instance, certain tetrahydroisoquinoline derivatives have been investigated for their potential therapeutic effects in neurological conditions. citeturn0search9 Due to its complex structure and the presence of multiple functional groups, (S)-4-(3-Bromophenyl)-6,8-dichloro-2-methyl-1,2,3,4-tetrahydroisoquinoline may serve as an intermediate in organic synthesis, particularly in the development of compounds with potential biological activity. However, specific applications of this compound have not been extensively documented in the available scientific literature. References none |
| Market Analysis Reports |