Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Classification | Flavors and spices >> Synthetic spice >> Lactone and oxygen-containing heterocyclic compound >> Furan and pyran |
|---|---|
| Name | 4-(4-Bromophenyl)dibenzofuran |
| Molecular Structure | 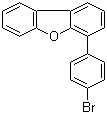 |
| Molecular Formula | C18H11BrO |
| Molecular Weight | 323.18 |
| CAS Registry Number | 955959-84-9 |
| EC Number | 815-536-8 |
| SMILES | C1=CC=C2C(=C1)C3=C(O2)C(=CC=C3)C4=CC=C(C=C4)Br |
| Solubility | Insoluble (2.7E-6 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.456±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Index of Refraction | 1.708, Calc.* |
| Boiling Point | 460.1±20.0 °C (760 mmHg), Calc.* |
| Flash Point | 232.1±21.8 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H335 Details | ||||||||||||
| Safety Statements | P261-P264-P271-P280-P302+P352-P304+P340-P319-P321-P332+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
4-(4-Bromophenyl)dibenzofuran is an organic compound belonging to the class of dibenzofurans. It is characterized by a dibenzofuran structure with a bromophenyl group attached at the 4-position of one of the phenyl rings. This compound has gained interest primarily due to its potential applications in materials science, specifically in the development of organic electronic materials and as an intermediate in the synthesis of other complex organic compounds. The discovery and development of 4-(4-bromophenyl)dibenzofuran can be traced to the broader research into dibenzofuran derivatives. Dibenzofuran itself is a heterocyclic compound composed of two fused benzene rings with an oxygen atom bridging them. Its derivatives, such as 4-(4-bromophenyl)dibenzofuran, exhibit a wide range of properties that make them valuable in various fields, including organic electronics and pharmaceutical synthesis. The compound is generally synthesized via standard synthetic methods for functionalized dibenzofurans. One common route involves the Friedel-Crafts acylation reaction, where a bromophenyl group is introduced to a dibenzofuran precursor. This synthesis is widely utilized and has been well documented in the chemical literature. The ability to selectively place substituents such as the bromophenyl group at specific positions on the dibenzofuran ring system allows for tuning of the compound’s electronic and steric properties. In terms of applications, 4-(4-bromophenyl)dibenzofuran is primarily studied for its potential use in organic semiconductors, particularly in the field of organic light-emitting diodes (OLEDs) and organic solar cells (OPVs). The introduction of the bromophenyl group into the dibenzofuran structure can significantly affect the electronic properties of the compound, such as its charge transport characteristics, which are crucial for its performance in organic electronic devices. Its use as a building block in the synthesis of more complex organic semiconductors is of particular interest in the ongoing development of organic electronic materials. Additionally, 4-(4-bromophenyl)dibenzofuran has been investigated for its potential use in the synthesis of bioactive molecules. Due to its structure, it can serve as a versatile intermediate for creating more complex compounds that may have pharmacological activity or serve as molecular probes in biological research. The bromophenyl group can also participate in various cross-coupling reactions, such as Suzuki or Heck reactions, to introduce further functional groups, expanding the range of possible applications in both material science and medicinal chemistry. The compound's role in organic electronics and synthetic chemistry highlights its importance as a structural unit for the design and development of advanced materials. Its synthesis and functionalization have been well-established, and ongoing research continues to explore new methods of utilizing 4-(4-bromophenyl)dibenzofuran in the creation of next-generation electronic devices and pharmaceuticals. In conclusion, 4-(4-bromophenyl)dibenzofuran is an important compound with applications in organic electronics, material science, and synthetic chemistry. Its unique structure and functional properties make it a valuable building block for the development of advanced materials and bioactive molecules, contributing to both industrial and academic advancements. References 2022. Current State, Challenges, and Perspectives on Microbial Degradation of Dioxin and Furan. Enzymes for Pollutant Degradation. DOI: 10.1007/978-981-16-4574-7_13 |
| Market Analysis Reports |