Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Beijing Eagle Sky Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.eagleskypharmatech.com | |||
 | +86 (10) 5979-9429 8875-5821 | |||
 | +86 (10) 5804-3698 | |||
 | sophia_818@126.com contact@eagleskypharmatech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Hongsheng Industry Limited Company | China | |||
|---|---|---|---|---|
 | www.shhschem.net | |||
 | +86 (21) 5426-5267 +86 13901708303 | |||
 | +86 (21) 5426-5268 | |||
 | chshhsc@online.sh.cn zhgshhs@hotmail.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1998 | ||||
| chemBlink Standard supplier since 2012 | ||||
| Beijing Lang Rebone Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.langrb.com | |||
 | +86 (10) 5260-9649 +86 18612220856 | |||
 | +86 (10) 8895-6459 | |||
 | sales@langrb.com marketing@langrb.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Nanjing Bic Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.bicbiotech.com | |||
 | +86 18851183712 | |||
 | sales@bicbiotech.com | |||
 | QQ Chat | |||
 | WeChat: +8618851183712 | |||
 | WhatsApp:+8618851183712 | |||
| Chemical manufacturer since 2023 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Dalton Pharma Services | Canada | |||
|---|---|---|---|---|
 | www.dalton.com | |||
 | +1 (416) 661-2102 | |||
 | +1 (416) 661-2108 | |||
 | chemist@dalton.com | |||
| Chemical manufacturer | ||||
| Carbone Scientific Co., Ltd. | UK | |||
|---|---|---|---|---|
 | www.carbonesci.com | |||
 | +44 (870) 486-8629 | |||
 | +44 (870) 288-7399 | |||
 | sales@carbonesci.com | |||
| Chemical distributor | ||||
| Eburon Organics bvba | Belgium | |||
|---|---|---|---|---|
 | www.eburon-organics.com | |||
 | +32 (51) 335-068 | |||
 | +32 (51) 335-064 | |||
 | sales@eburon-organics.com | |||
| Chemical manufacturer | ||||
| Ryan Scientific, Inc. | USA | |||
|---|---|---|---|---|
 | www.ryansci.com | |||
 | +1 (843)-884-4911 | |||
 | +1 (843) 884-5568 | |||
 | sales@ryansci.com | |||
| Chemical manufacturer | ||||
| Classification | Chemical reagent >> Organic reagent >> Fatty alcohol |
|---|---|
| Name | Triethylene glycol di(p-toluenesulfonate) |
| Synonyms | 2-[2-[2-(4-methylphenyl)sulfonyloxyethoxy]ethoxy]ethyl 4-methylbenzenesulfonate |
| Molecular Structure | 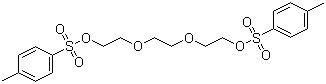 |
| Molecular Formula | C20H26O8S2 |
| Molecular Weight | 458.55 |
| CAS Registry Number | 19249-03-7 |
| EC Number | 628-844-0 |
| SMILES | CC1=CC=C(C=C1)S(=O)(=O)OCCOCCOCCOS(=O)(=O)C2=CC=C(C=C2)C |
| Density | 1.3$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Melting point | 78 - 82 $degree$C (Expl.) |
| Boiling point | 608.4$+/-$55.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 321.7$+/-$31.5 $degree$C (Calc.)* |
| Index of refraction | 1.545 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Triethylene glycol di(p-toluenesulfonate) is an organic compound belonging to the class of diesters derived from sulfonic acids and polyether alcohols. It is formed by the esterification of triethylene glycol, a linear polyether with three ethylene oxide units, with p-toluenesulfonyl chloride (tosyl chloride) to produce the corresponding di-p-toluenesulfonate. Compounds of this type are commonly used as activated intermediates in organic synthesis due to the excellent leaving-group ability of the tosylate group, which facilitates nucleophilic substitution reactions. The preparation of triethylene glycol di(p-toluenesulfonate) involves reacting triethylene glycol with two equivalents of tosyl chloride in the presence of a base, such as pyridine or triethylamine, to neutralize the hydrochloric acid byproduct. The resulting compound contains two tosylate ester groups at the terminal hydroxyl positions of the glycol chain, which are highly reactive toward nucleophiles, allowing for efficient conversion into ethers, thioethers, or other functionalized derivatives. The linear triethylene glycol backbone provides flexibility and hydrophilicity, making it suitable for use as a linker in multi-step synthetic pathways or as a spacer in polymer and bioconjugation chemistry. Triethylene glycol di(p-toluenesulfonate) has been widely used in the synthesis of oligoether derivatives, polyethylene glycol (PEG) analogs, and macrocyclic compounds. In nucleophilic substitution reactions, the tosylate groups can be displaced by halides, azides, thiols, or amines to form a wide variety of functionalized products. This reactivity is exploited in both small-molecule synthesis and polymer chemistry, where the glycol chain provides solubility and flexibility, and the tosylate groups act as reactive handles for further functionalization. In medicinal chemistry, triethylene glycol di(p-toluenesulfonate) is employed as an intermediate in the preparation of prodrugs, PEGylated conjugates, and other compounds where a hydrophilic linker is desirable. The di-tosylate can be reacted with nucleophilic drug molecules or peptides to form stable ether linkages, improving solubility, pharmacokinetics, and bioavailability. The ability to selectively functionalize each terminal position of the glycol chain allows for controlled synthesis of asymmetrically substituted derivatives, which can be important in targeted drug delivery or bioconjugation. In addition to its use in synthetic and medicinal chemistry, this compound finds applications in materials science. The glycol backbone imparts flexibility, while the tosylate groups allow incorporation into polymer networks, cross-linked materials, and surface-functionalized polymers. Such modifications can enhance polymer properties, including solubility, thermal stability, and reactivity for post-polymerization functionalization. Overall, triethylene glycol di(p-toluenesulfonate) serves as a versatile activated diester intermediate in organic chemistry. Its combination of a flexible triethylene glycol backbone and highly reactive tosylate groups enables its use in nucleophilic substitution reactions, polymer functionalization, bioconjugation, and prodrug synthesis. This compound illustrates the utility of sulfonate esters as leaving groups and the importance of polyether linkers in modern synthetic methodologies. References 2023. Ion valence-gated photochromism of an aza-crowned diarylethene. Photochemical & Photobiological Sciences. DOI: 10.1007/s43630-023-00508-z 2021. Synthesis and properties of heterocycle-containing podands. Chemistry of Heterocyclic Compounds. DOI: 10.1007/s10593-021-03009-y |
| Market Analysis Reports |