Online Database of Chemicals from Around the World
| Matrix Scientific Inc. | USA | |||
|---|---|---|---|---|
 | www.matrixscientific.com | |||
 | +1 (803) 788-9494 | |||
 | +1 (803) 788-9419 | |||
 | sales@matrixscientific.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Aryl compounds >> Naphthalenes |
|---|---|
| Name | 7-(Dimethoxymethyl)-1,2,3,4-tetrahydro-1,8-naphthyridine |
| Molecular Structure | 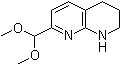 |
| Molecular Formula | C11H16N2O2 |
| Molecular Weight | 208.26 |
| CAS Registry Number | 204452-91-5 |
| SMILES | COC(C1=NC2=C(CCCN2)C=C1)OC |
| Solubility | Very slightly soluble (0.25 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.20±0.1 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 91-92.5 °C** |
| Boiling point | 311.0±42.0 °C 760 mmHg (Calc.)* |
| Flash point | 141.9±27.9 °C (Calc.)* |
| Index of refraction | 1.523 (Calc.)*16.2 °C (Calc.)* |
| Index of refraction | 1.586 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2014 ACD/Labs) |
| ** | Yasuda, Nobuyoshi |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302 Details |
| Safety Statements | P280-P305+P351+P338 Details |
| SDS | Available |
|
7-(Dimethoxymethyl)-1,2,3,4-tetrahydro-1,8-naphthyridine is a chemical compound that belongs to the class of tetrahydronaphthyridine derivatives, which feature a bicyclic structure containing both nitrogen and carbon atoms. The compound is characterized by the presence of a dimethoxymethyl group at the 7-position of the naphthyridine ring, a feature that influences its reactivity and potential applications in synthetic chemistry and pharmaceutical research. The discovery of 7-(dimethoxymethyl)-1,2,3,4-tetrahydro-1,8-naphthyridine is part of ongoing efforts to explore the chemical space of heterocyclic compounds, particularly those involving naphthyridine, a well-studied scaffold in medicinal chemistry. Naphthyridine derivatives are known for their broad range of biological activities, including antimicrobial, antiviral, anticancer, and anti-inflammatory effects. The specific functionalization of the 7-position with a dimethoxymethyl group provides an opportunity to enhance the chemical stability of the molecule while also enabling selective modifications under specific conditions. The dimethoxymethyl group in this compound is significant because it acts as a protecting group for a reactive functional group, such as an aldehyde or alcohol. This feature is crucial in synthetic chemistry, where protection and deprotection steps are common strategies used to control reactivity during multi-step organic syntheses. In particular, the dimethoxymethyl group can be cleaved under acidic conditions to yield a corresponding aldehyde or alcohol, allowing for further functionalization of the naphthyridine ring. This type of selective manipulation is highly valuable for the preparation of more complex molecules with tailored properties. As a synthetic intermediate, 7-(dimethoxymethyl)-1,2,3,4-tetrahydro-1,8-naphthyridine can be utilized in the construction of a variety of other naphthyridine derivatives. These derivatives may possess enhanced biological activity or target specific molecular pathways involved in disease processes. For instance, modifications to the naphthyridine core can result in compounds that exhibit potent interactions with DNA or RNA, making them potential candidates for use in anti-cancer or anti-viral therapies. The ability to modify the structure of the compound through reactions such as cyclization, cross-coupling, or condensation is a key reason for its value in pharmaceutical development. One area of research where naphthyridine derivatives, including 7-(dimethoxymethyl)-1,2,3,4-tetrahydro-1,8-naphthyridine, have been explored is in the design of small molecule inhibitors of important enzymes. For example, these compounds can inhibit the activity of kinases, proteases, or other enzymes that play crucial roles in cellular signaling and disease progression. By attaching various substituents at specific positions on the naphthyridine ring, researchers can modulate the molecule’s binding affinity, specificity, and stability, which are essential properties for drug development. Furthermore, tetrahydronaphthyridine derivatives like 7-(dimethoxymethyl)-1,2,3,4-tetrahydro-1,8-naphthyridine have also been studied for their potential neuroactive properties. The tetrahydropyridine substructure is often found in compounds that affect neurotransmission and neuronal activity. These molecules may interact with receptors or enzymes in the brain, making them candidates for the development of drugs aimed at treating neurological conditions such as Alzheimer’s disease, Parkinson’s disease, or depression. The versatility of 7-(dimethoxymethyl)-1,2,3,4-tetrahydro-1,8-naphthyridine in organic synthesis and medicinal chemistry arises from its potential to undergo a variety of chemical transformations. Through controlled reactions, such as the selective removal of the dimethoxymethyl group, further functional groups can be introduced, allowing for the synthesis of complex molecules with targeted pharmacological profiles. These transformations make the compound a valuable intermediate in the synthesis of biologically active naphthyridine derivatives. In conclusion, 7-(dimethoxymethyl)-1,2,3,4-tetrahydro-1,8-naphthyridine is a useful chemical intermediate in synthetic organic chemistry, particularly in the design and development of novel pharmaceutical agents. Its structure offers a platform for the development of compounds with a range of biological activities, including anticancer, antimicrobial, antiviral, and neuroactive effects. The ability to modify the compound through selective reactions makes it an important tool in the exploration of new therapeutic agents. |
| Market Analysis Reports |