Online Database of Chemicals from Around the World
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Intatrade Chemicals GmbH | Germany | |||
|---|---|---|---|---|
 | www.intatrade.de | |||
 | +49 (3493) 605-465 | |||
 | +49 (3493) 605-470 | |||
 | sales@intatrade.de | |||
| Chemical distributor | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Changzhou Hongyu Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.czhongyu-chem.com | |||
 | +86 (519) 8999-6110 | |||
 | +86 (519) 8999-6110 | |||
 | 545701991@qq.com | |||
| Chemical distributor since 2011 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Epsilon Chimie Chemical Manufacturer | France | |||
|---|---|---|---|---|
 | www.epsilon-chimie.com | |||
 | +33 (2) 9842-4650 | |||
 | +33 (2) 9842-4402 | |||
 | pierre.cornec@epsilon-chimie.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | (Triphenylphosphoranylidene)acetaldehyde |
| Synonyms | Formylmethylene triphenylphosphorane |
| Molecular Structure | 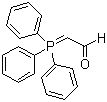 |
| Molecular Formula | C20H17OP |
| Molecular Weight | 304.32 |
| CAS Registry Number | 2136-75-6 |
| EC Number | 218-375-6 |
| SMILES | C1=CC=C(C=C1)P(=CC=O)(C2=CC=CC=C2)C3=CC=CC=C3 |
| Melting point | 185-188 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
(Triphenylphosphoranylidene)acetaldehyde is a notable compound in the field of organophosphorus chemistry, distinguished by its unique structure and versatile applications. This compound consists of a triphenylphosphoranylidene group attached to an acetaldehyde moiety, forming a molecule with significant chemical reactivity and utility in various chemical processes. The discovery of (triphenylphosphoranylidene)acetaldehyde emerged from the exploration of phosphoranylidene derivatives and their applications in organic synthesis. The synthesis of this compound typically involves the reaction of triphenylphosphine with acetaldehyde under controlled conditions to form the phosphoranylidene derivative. The resulting product features a phosphorus atom double-bonded to a carbonyl group and bonded to three phenyl rings, which imparts unique electronic and steric properties to the molecule. One of the key applications of (triphenylphosphoranylidene)acetaldehyde lies in its role as a reagent and intermediate in organic synthesis. The compound is used in various reactions, including the formation of phosphoranylidene derivatives, which are valuable in the synthesis of complex organic molecules. Its reactivity as a phosphoranylidene allows it to participate in condensation reactions, offering a pathway to synthesize a range of phosphorus-containing compounds. In addition to its role in organic synthesis, (triphenylphosphoranylidene)acetaldehyde is utilized in the development of novel materials and chemical processes. The compound's ability to form stable complexes with other reagents and catalysts makes it useful in the design of new materials with specific properties. For example, its interaction with metal centers can lead to the formation of organometallic compounds with potential applications in catalysis, materials science, and electronics. The study of (triphenylphosphoranylidene)acetaldehyde also contributes to the broader understanding of phosphorus chemistry and the design of phosphoranylidene derivatives. Researchers explore the properties of this compound to gain insights into the behavior of phosphoranylidene groups in various chemical contexts. This knowledge is valuable for developing new reagents and catalysts with enhanced performance and selectivity. Overall, (triphenylphosphoranylidene)acetaldehyde represents an important compound in organophosphorus chemistry, with applications in organic synthesis, materials development, and chemical research. Its unique structure and reactivity make it a valuable tool for advancing both fundamental chemistry and practical applications. References 2007. Wittig Reaction. Science of Synthesis. URL: SD-025-00553 2019. Recent advances in the applications of Wittig reaction in the total synthesis of natural products containing lactone, pyrone, and lactam as a scaffold. Monatshefte für Chemie - Chemical Monthly, 150(8). DOI: 10.1007/s00706-019-02465-9 2012. A practical route to long-chain non-natural α,ω-diamino acids. Amino Acids, 43(5). DOI: 10.1007/s00726-012-1349-0 |
| Market Analysis Reports |