Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Pyrimidine compound >> Ketones |
|---|---|
| Name | 10-Ethyl-9(10H)-acridinone |
| Synonyms | 10-Ethyl-9-Acridone; 10-Ethyl-9-acridanone; N-Ethylacridone; NSC 117669 |
| Molecular Structure | 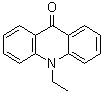 |
| Molecular Formula | C15H13NO |
| Molecular Weight | 223.27 |
| CAS Registry Number | 2207-41-2 |
| SMILES | CCN1C2=CC=CC=C2C(=O)C3=CC=CC=C31 |
| Solubility | Insoluble (8.9E-4 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.167±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 162.5-163.0 °C** (Expl.) |
| Index of Refraction | 1.616, Calc.* |
| Boiling Point | 373.5±12.0 °C (760 mmHg), Calc.* |
| Flash Point | 148.2±8.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2019 ACD/Labs) |
| ** | Akiba, Kinya |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details |
|---|---|
| Risk Statements | H302 Details |
| Safety Statements | P264-P270-P301+P312-P330-P501 Details |
| SDS | Available |
|
10-Ethyl-9(10H)-acridinone is a chemical compound belonging to the acridine family, a group of heterocyclic compounds featuring a fused benzene and pyridine ring system. This particular compound is derived by introducing an ethyl group at the 10-position of the acridinone structure, which significantly alters its chemical and physical properties compared to the parent compound, 9(10H)-acridinone. The discovery of 10-ethyl-9(10H)-acridinone is rooted in the ongoing exploration of acridine derivatives for their diverse biological and chemical activities. Acridines and their derivatives have been extensively studied since the mid-20th century, primarily due to their applications in areas such as medicinal chemistry, organic electronics, and materials science. The introduction of an ethyl group to the 10-position of the acridinone ring system was hypothesized to enhance the solubility of the compound in organic solvents while preserving its photophysical properties, making it a promising candidate for use in a variety of applications. One of the key applications of 10-ethyl-9(10H)-acridinone lies in the field of medicinal chemistry. Acridines are well known for their antitumor and antimicrobial properties, and derivatives like 10-ethyl-9(10H)-acridinone are studied for their potential use in cancer therapy. The compound's ability to intercalate into DNA has been recognized as a mechanism for inhibiting cell division, which is a critical feature for anticancer agents. Research into the modification of acridines with alkyl groups, such as the ethyl group at the 10-position, seeks to improve the compound's pharmacokinetic properties, such as solubility and stability, while maintaining its bioactivity. This makes 10-ethyl-9(10H)-acridinone a potential lead compound for further development as an anticancer drug. In addition to its potential medicinal applications, 10-ethyl-9(10H)-acridinone has been explored for its photophysical properties. The compound exhibits fluorescence, which makes it useful in the development of fluorescent probes for biological imaging. Such probes can be utilized in various research fields, including the study of cellular processes and the detection of biomolecules. The fluorescence of 10-ethyl-9(10H)-acridinone is sensitive to the environment, and its emission properties can be tuned by altering the substituents on the acridine ring, making it a versatile tool for bioanalytical applications. In summary, 10-ethyl-9(10H)-acridinone is a promising compound with applications in medicinal chemistry, particularly in cancer research, as well as in the development of fluorescent probes for biological imaging. Its ability to intercalate into DNA and exhibit fluorescence makes it a valuable candidate for further study and potential therapeutic use. References 1976. Reaction of N-heteroaromatic cations with alkali-metal amides. Chemistry of Heterocyclic Compounds, 12(3). DOI: 10.1007/bf00479570 2023. Multimodal action of KRP203 on phosphoinositide kinases in vitro and in cells. Biochemical and Biophysical Research Communications, 682. DOI: 10.1016/j.bbrc.2023.08.050 2003. Melting, volatilisation and crystal lattice enthalpies of acridin-9(10H)-ones. Journal of Thermal Analysis and Calorimetry, 74(2). DOI: 10.1023/b:jtan.0000005179.91819.6d |
| Market Analysis Reports |