Online Database of Chemicals from Around the World
| Wilshire Technologies, Inc. | USA | |||
|---|---|---|---|---|
 | www.wilshiretechnologies.com | |||
 | +1 (609) 683-1117 | |||
 | +1 (732) 274-0049 | |||
 | Wilshire-info@evonik.com | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Shanghai Taoe Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.taoe-chem.com | |||
 | +86 (21) 5153-1816 | |||
 | +86 (21) 5153-1817 | |||
 | sales@taoe-chem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2014 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Yantai Xianhua Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.xianhualc.com | |||
 | +86 (535) 693-4138 +86 18663898285 | |||
 | +86 (535) 693-4137 | |||
 | sales@xianhualc.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2003 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zhejiang Free Trade Zone Violet Trading Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zjviolettech.com | |||
 | +86 (580) 811-8992 | |||
 | +86 (580) 811-9487 | |||
 | jenny@zjviolettech.com | |||
| Chemical distributor since 2019 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Henan Ouber Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oubertec.com | |||
 | +86 (371) 6532-2607 +86 18937141980 | |||
 | +86 (371) 6532-2607 | |||
 | anna.zhang@oubertec.com | |||
 | QQ Chat | |||
 | WeChat: 18937141980 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| HanChem Co., Ltd. Head Office | South Korea | |||
|---|---|---|---|---|
 | www.hanchem.net | |||
 | +82 (42) 931-2640 | |||
 | +82 (42) 931-2643 | |||
 | sales@hanchem.net | |||
| Chemical manufacturer since 1999 | ||||
| AK Scientific, Inc | USA | |||
|---|---|---|---|---|
 | www.aksci.com | |||
 | +1 (510) 429-8835 | |||
 | +1 (510) 429-8836 | |||
 | sales@aksci.com | |||
| Chemical manufacturer | ||||
| Classification | Pharmaceutical intermediate >> Heterocyclic compound intermediate >> Thiophene compound |
|---|---|
| Name | 2-Bromodibenzothiophene |
| Synonyms | 2-Bromodibenzothiophene; NSC 172586 |
| Molecular Structure | 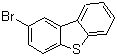 |
| Molecular Formula | C12H7BrS |
| Molecular Weight | 263.15 |
| CAS Registry Number | 22439-61-8 |
| EC Number | 891-396-1 |
| SMILES | C1=CC=C2C(=C1)C3=C(S2)C=CC(=C3)Br |
| Density | 1.611 |
|---|---|
| Melting point | 250-252 °C |
| Boiling point | 387 °C |
| Flash point | 188 °C |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
2-Bromodibenzothiophene is a notable chemical compound with significant implications in both synthetic chemistry and material science. Its discovery and applications have highlighted its value in various chemical processes, particularly in the development of advanced materials and pharmaceuticals. The compound 2-Bromodibenzothiophene was first identified in the late 20th century as part of research efforts to explore new heterocyclic compounds with potential applications in organic synthesis. Dibenzothiophene itself is a well-known structure in organic chemistry, and its brominated derivative has been studied for its enhanced reactivity and utility in various chemical transformations. The introduction of the bromine atom at the 2-position of the dibenzothiophene ring system significantly alters its chemical properties, making it a valuable intermediate in a range of chemical processes. The synthesis of 2-Bromodibenzothiophene typically involves the bromination of dibenzothiophene. This process is generally carried out using bromine or brominating agents in the presence of a suitable solvent. The reaction conditions are carefully controlled to ensure selective bromination at the desired position while avoiding overbromination or other side reactions. The resulting product is purified through standard techniques such as recrystallization or chromatography to obtain the high-purity compound required for further applications. One of the primary applications of 2-Bromodibenzothiophene is in the field of organic synthesis. The bromine atom in this compound acts as a versatile functional group, facilitating various chemical reactions, including coupling reactions and nucleophilic substitutions. For example, 2-Bromodibenzothiophene can be used as a building block in the synthesis of more complex molecules, including pharmaceuticals and organic materials. Its reactivity makes it a valuable intermediate for constructing various chemical architectures. In addition to its role in organic synthesis, 2-Bromodibenzothiophene is also used in the development of advanced materials. The compound's unique structural features and reactivity make it suitable for applications in organic electronics and optoelectronics. For instance, it can be used as a precursor in the synthesis of organic semiconductors and conducting polymers, which are essential components in electronic devices such as organic light-emitting diodes (OLEDs) and organic photovoltaic cells. The presence of the bromine atom in 2-Bromodibenzothiophene can influence the electronic properties of the resulting materials, enhancing their performance in various applications. The advantages of using 2-Bromodibenzothiophene in these applications include its ability to introduce specific functional groups into chemical structures and its contribution to the development of advanced materials with tailored properties. However, there are challenges associated with its use, such as the need for careful handling and control of reaction conditions to avoid unwanted side reactions. Ongoing research aims to optimize the synthesis and application of 2-Bromodibenzothiophene, addressing these challenges and exploring new uses for this compound. Future research directions for 2-Bromodibenzothiophene may focus on expanding its applications in new areas of organic synthesis and material science. Researchers may explore its potential in developing new types of electronic materials or in designing innovative pharmaceuticals with improved properties. Advances in these areas could further enhance the utility of 2-Bromodibenzothiophene and contribute to the development of new technologies and products. References 2024. Exploring the effects of mono-bromination on hole-electron transport and distribution in dibenzofuran and dibenzothiophene isomers: a first-principles study. *Journal of Molecular Modeling*, 30(6). DOI: 10.1007/s00894-024-05966-5 2021. Different molecular conformation and packing determining mechanochromism and room-temperature phosphorescence. *Science China Materials*, 64(5). DOI: 10.1007/s40843-021-1658-9 2001. Halogenation. *Science of Synthesis*. URL: SD-010-00337 |
| Market Analysis Reports |