Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Molcore Biopharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.molcore.com | |||
 | +86 (571) 8102-5280 | |||
 | sales@molcore.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Xinxiang Runyu Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runvmat.com | |||
 | +86 (373) 775-9608 | |||
 | +86 (373) 775-9608 | |||
 | sales@runvmat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Henan Ouber Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.oubertec.com | |||
 | +86 (371) 6532-2607 +86 18937141980 | |||
 | +86 (371) 6532-2607 | |||
 | anna.zhang@oubertec.com | |||
 | QQ Chat | |||
 | WeChat: 18937141980 | |||
| Chemical manufacturer since 2020 | ||||
| chemBlink Standard supplier since 2020 | ||||
| Anhui Sholon New Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sholonchem.com | |||
 | +86 (0550) 526-1666 | |||
 | +86 (550) 526-1555 | |||
 | info@sholonchem.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Shanghai Worldyang Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.worldyachem.com | |||
 | +86 13651600618 +86 (21) 5679-5779 | |||
 | +86 (21) 5679-5266 | |||
 | sales7777@worldyachem.com | |||
 | QQ Chat | |||
 | WeChat: 13651600618 | |||
 | WhatsApp:+86 13651600618 | |||
| Chemical manufacturer since 2012 | ||||
| Classification | Chemical reagent >> Organic reagent >> Tricyclic compound |
|---|---|
| Name | 1-Bromodibenzofuran |
| Molecular Structure | 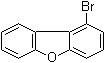 |
| Molecular Formula | C12H7BrO |
| Molecular Weight | 247.09 |
| CAS Registry Number | 50548-45-3 |
| EC Number | 837-464-6 |
| SMILES | C1=CC=C2C(=C1)C3=C(O2)C=CC=C3Br |
| Solubility | Insoluble (1.2E-4 g/L) (25 °C), Calc.* |
|---|---|
| Density | 1.577±0.06 g/cm3 (20 °C 760 Torr), Calc.* |
| Melting point | 67 °C** |
| Index of Refraction | 1.721, Calc.* |
| Boiling Point | 343.8±15.0 °C (760 mmHg), Calc.* |
| Flash Point | 161.7±20.4 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software V11.02 (©1994-2014 ACD/Labs) |
| ** | Van Ess, Paul Raymond |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319 Details | ||||||||||||
| Safety Statements | P264-P264+P265-P280-P302+P352-P305+P351+P338-P321-P332+P317-P337+P317-P362+P364 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
1-Bromodibenzofuran is an organic compound that belongs to the class of dibenzofuran derivatives. It features a bromine atom substituted at the 1-position of the dibenzofuran ring. The molecular formula of 1-bromodibenzofuran is C12H7BrO, with the structure comprising two benzene rings fused to an oxygen-containing heterocyclic ring. Dibenzofuran derivatives, such as 1-bromodibenzofuran, have been studied for their chemical properties, reactivity, and potential applications in various fields, including organic synthesis and materials science. The discovery of dibenzofuran and its derivatives, including 1-bromodibenzofuran, is linked to early studies of polycyclic aromatic compounds, which gained attention due to their unique chemical properties and structural complexity. The specific introduction of a bromine atom to the 1-position of the dibenzofuran ring is typically achieved through a bromination reaction using bromine or bromine-based reagents. This reaction selectively replaces a hydrogen atom on the aromatic ring, resulting in the formation of the 1-bromodibenzofuran derivative. 1-Bromodibenzofuran is primarily used as an intermediate in organic synthesis. Its structure makes it an important building block for the construction of more complex molecules. Due to its reactivity, 1-bromodibenzofuran can undergo various chemical reactions, especially electrophilic substitution reactions. These reactions allow the selective introduction of functional groups into the dibenzofuran ring, which can be useful in the preparation of pharmaceuticals, agrochemicals, and other specialty chemicals. In addition to its use in organic synthesis, 1-bromodibenzofuran has potential applications in materials science, particularly in the field of organic electronics. Dibenzofuran and its derivatives have been explored for their ability to function as organic semiconductors in optoelectronic devices. The presence of the bromine atom at the 1-position of the dibenzofuran ring can influence the electronic properties of the compound, making it potentially useful in the development of organic light-emitting diodes (OLEDs), organic solar cells (OPVs), and organic field-effect transistors (OFETs). The electronic properties of dibenzofuran derivatives are of interest because of their ability to facilitate charge transport, an important characteristic for materials used in organic electronics. The synthesis of 1-bromodibenzofuran, as well as other halogenated dibenzofuran derivatives, provides a pathway for the creation of functionalized compounds that can be further modified to tailor their properties for specific applications. The introduction of the bromine atom can also affect the stability and reactivity of the compound, allowing it to be used as a precursor for the synthesis of various other molecules. This versatility makes 1-bromodibenzofuran a valuable intermediate in the development of new chemical compounds for diverse industrial and scientific purposes. While 1-bromodibenzofuran has not been widely studied for direct biological applications, the compound's role as a precursor in the synthesis of other functionalized aromatic compounds holds promise. Many polycyclic aromatic compounds, including dibenzofuran derivatives, have been investigated for their potential biological activities, such as antimicrobial, anticancer, and antioxidant properties. The ability to modify the dibenzofuran structure through the introduction of various substituents, including halogens like bromine, may result in compounds with distinct biological properties. In conclusion, 1-bromodibenzofuran is a chemically significant compound that serves primarily as an intermediate in organic synthesis and materials science. Its role as a precursor for the synthesis of complex molecules and its potential applications in the development of organic semiconductors highlight its value in various scientific and industrial fields. As with other dibenzofuran derivatives, the compound's versatility and reactivity make it an important building block for the design and development of new materials and chemical entities. References 1995. Polybrominated Biphenyl and Diphenylether Flame Retardants: Analysis, Toxicity, and Environmental Occurrence. Reviews of Environmental Contamination and Toxicology, 141. DOI: 10.1007/978-1-4612-2530-0_1 2016. Estimation of melting points of large set of persistent organic pollutants utilizing QSPR approach. Journal of Molecular Modeling, 22(3). DOI: 10.1007/s00894-016-2917-0 2024. Exploring the effects of mono-bromination on hole-electron transport and distribution in dibenzofuran and dibenzothiophene isomers: a first-principles study. Journal of Molecular Modeling, 30(6). DOI: 10.1007/s00894-024-05966-5 |
| Market Analysis Reports |