Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hangzhou Brown Bio-Pharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chinaelion.com | |||
 | +86 (571) 8538-1539 +86 13588363288 | |||
 | +86 (571) 8538-1539 / 8502-2151 | |||
 | elionchem@chinaelion.com sales@chinaelion.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2013 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Biosynth AG. | Switzerland | |||
|---|---|---|---|---|
 | www.biosynth.com | |||
 | +41 (71) 858-2020 | |||
 | +41 (71) 858-2030 | |||
 | welcome@biosynth.ch | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2014 | ||||
| Classification | Chemical reagent >> Organic reagent >> Cyanate ester / isocyanate |
|---|---|
| Name | Triphenylmethane-4,4',4''-triisocyanate |
| Synonyms | Tris(4-isocyanatophenyl)methane |
| Molecular Structure | 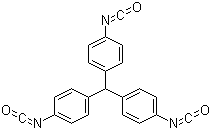 |
| Molecular Formula | C22H13N3O3 |
| Molecular Weight | 367.36 |
| CAS Registry Number | 2422-91-5 |
| EC Number | 219-351-8 |
| SMILES | C1=CC(=CC=C1C(C2=CC=C(C=C2)N=C=O)C3=CC=C(C=C3)N=C=O)N=C=O |
| Density | 1.2±0.1 g/cm3, Calc.*, 1.19 g/mL |
|---|---|
| Index of Refraction | 1.621, Calc.* |
| Boiling Point | 498.5±45.0 °C (760 mmHg), Calc.* |
| Flash Point | 204.7±34.1 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS06;GHS07;GHS08 Danger Details GHS06;GHS07;GHS08 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H317-H319-H330-H334-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P233-P260-P261-P264-P264+P265-P270-P271-P272-P280-P284-P301+P317-P302+P352-P304+P340-P305+P351+P338-P316-P319-P320-P321-P330-P332+P317-P333+P317-P337+P317-P342+P316-P362+P364-P403-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
|
Triphenylmethane-4,4',4''-triisocyanate, commonly known as TPTI, is a significant isocyanate compound that has gained attention for its unique chemical properties and diverse applications in various industries. This compound is a member of the triphenylmethane family, characterized by a central carbon atom bonded to three phenyl groups and three isocyanate functional groups. The development and discovery of TPTI date back to the advancements in organic chemistry during the mid-20th century, particularly in the context of isocyanate chemistry and its applications in polymer science. The synthesis of triphenylmethane-4,4',4''-triisocyanate typically involves the reaction of triphenylmethanol with phosgene or other isocyanate-producing agents. The resulting compound features a highly reactive isocyanate functional group, which can undergo various chemical reactions, including polymerization and cross-linking with polyols. This reactivity is one of the key characteristics that make TPTI valuable in the production of polyurethanes and other advanced polymeric materials. One of the primary applications of triphenylmethane-4,4',4''-triisocyanate is in the manufacture of polyurethane foams, elastomers, and coatings. In these applications, TPTI acts as a hardener or cross-linking agent, promoting the reaction between isocyanates and polyols to form stable polymeric networks. The resulting polyurethanes exhibit excellent mechanical properties, durability, and resistance to heat, making them suitable for various industrial and consumer applications. For instance, TPTI-based polyurethane foams are commonly used in furniture, automotive interiors, insulation materials, and footwear, where lightweight yet strong materials are required. In addition to its role in polyurethanes, triphenylmethane-4,4',4''-triisocyanate is also utilized in the formulation of coatings and adhesives. Its ability to provide a strong adhesive bond and enhance the durability of coatings makes it a valuable additive in the paint and coatings industry. TPTI-based coatings are known for their excellent weather resistance, hardness, and chemical stability, making them ideal for outdoor applications such as protective coatings for buildings and infrastructure. Furthermore, TPTI has found applications in the field of specialty chemicals, particularly in the production of elastomers and thermoplastics. The incorporation of TPTI into polymer formulations allows for the tailoring of mechanical properties, such as flexibility, tensile strength, and tear resistance. This versatility enables manufacturers to develop products that meet specific performance criteria for a wide range of applications. Despite its advantages, the use of triphenylmethane-4,4',4''-triisocyanate poses certain health and safety concerns, primarily due to the toxicity associated with isocyanate compounds. Exposure to isocyanates can lead to respiratory issues and other health effects, necessitating the implementation of appropriate safety measures during handling and processing. As a result, industries are encouraged to adopt best practices for the safe use of TPTI and to explore alternatives that may offer similar performance with reduced health risks. In summary, triphenylmethane-4,4',4''-triisocyanate is an essential compound in the field of polymer chemistry, with significant applications in the production of polyurethanes, coatings, and specialty chemicals. Its discovery and utilization have contributed to advancements in material science, providing solutions for various industrial needs. As research continues to focus on improving safety and exploring alternative compounds, TPTI remains a critical player in the development of innovative polymeric materials. References 2016. Synthesis and properties of blocked Di- and polyisocyanates. *Polymer Science, Series D*, 9(2). DOI: 10.1134/s1995421216020064 2006. Plastic Materials. *Contact Dermatitis*. DOI: 10.1007/3-540-31301-x_34 2000. Polyurethane Resins. *Handbook of Occupational Dermatology*. DOI: 10.1007/978-3-662-07677-4_75 |
| Market Analysis Reports |