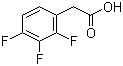
2,3,4-Trifluorophenylacetic acid is a chemical compound that belongs to the class of aromatic acids, specifically phenylacetic acids, with three fluorine atoms attached to the benzene ring in the 2, 3, and 4 positions. The presence of these fluorine atoms gives the compound unique chemical properties, which have made it useful in various applications, particularly in the synthesis of pharmaceuticals and agrochemicals.
The discovery and characterization of 2,3,4-trifluorophenylacetic acid have been driven by the need for new compounds with enhanced bioactivity, particularly those that exhibit improved stability or target specificity in pharmaceutical applications. The introduction of fluorine atoms into an aromatic ring typically influences the compound's electronic properties, such as its reactivity, hydrophobicity, and ability to interact with biological systems, making it a valuable building block in medicinal chemistry.
One of the main applications of 2,3,4-trifluorophenylacetic acid is in the field of pharmaceuticals. Fluorine substitution in organic molecules can modify their pharmacokinetic properties, such as bioavailability, metabolic stability, and binding affinity to biological targets. As a result, fluorine-containing compounds are often used in drug development to enhance the effectiveness of therapeutic agents. For instance, derivatives of 2,3,4-trifluorophenylacetic acid may be explored as intermediates for the synthesis of drugs that target various diseases, including cancer, cardiovascular diseases, and inflammatory disorders.
In agrochemicals, 2,3,4-trifluorophenylacetic acid and its derivatives are sometimes utilized in the development of herbicides or pesticides. Fluorine substitution can improve the potency and selectivity of these chemicals, allowing them to target specific plant species or pests while minimizing environmental impact. The ability to introduce trifluoromethyl groups into a molecule also enhances the compound's stability under environmental conditions, which is a valuable property for agrochemical formulations.
The chemical structure of 2,3,4-trifluorophenylacetic acid also makes it an interesting candidate for use in materials science. Fluorinated aromatic compounds often exhibit unique physical properties, such as increased resistance to heat and chemicals, as well as greater structural rigidity. These characteristics could potentially make 2,3,4-trifluorophenylacetic acid useful in the development of advanced materials for applications in electronics, coatings, and other high-performance materials.
Moreover, 2,3,4-trifluorophenylacetic acid could serve as an important intermediate in organic synthesis, particularly in the formation of more complex fluorinated compounds. By manipulating the carboxyl group or the trifluorophenyl moiety, synthetic chemists can prepare a variety of functionalized derivatives for use in further chemical reactions or as components in the design of new materials.
The introduction of fluorine atoms in 2,3,4-trifluorophenylacetic acid also influences its environmental behavior, particularly its solubility and persistence. Fluorine atoms can enhance a compound's resistance to biodegradation, making it more stable in various environmental conditions. This property can be beneficial in applications where long-term stability is required, but it also necessitates careful consideration of potential environmental impacts.
In summary, 2,3,4-trifluorophenylacetic acid is a fluorinated aromatic acid that finds applications in the synthesis of pharmaceuticals, agrochemicals, and advanced materials. Its unique chemical properties, including increased stability and the ability to modify biological interactions, make it a valuable compound in various fields of chemical research and development. By continuing to explore its potential in drug development and other industries, 2,3,4-trifluorophenylacetic acid plays a key role in advancing modern chemistry and technology.
|
























 GHS07 Warning Details
GHS07 Warning Details