Online Database of Chemicals from Around the World
| Hangzhou Verychem Science And Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.verychem.com | |||
 | +86 (571) 8816-2785 +86 13606544505 | |||
 | +86 (571) 8816-2787 | |||
 | lucy@verychem.com | |||
| Chemical manufacturer since 2004 | ||||
| chemBlink Massive supplier since 2021 | ||||
| Classification | Organic raw materials >> Nitrile compound |
|---|---|
| Name | 1,2,3-Tris(2-cyanoethoxy)propane |
| Synonyms | 3-[2,3-bis(2-cyanoethoxy)propoxy]propanenitrile |
| Molecular Structure | 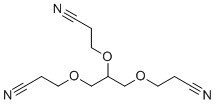 |
| Molecular Formula | C12H17N3O3 |
| Molecular Weight | 251.28 |
| CAS Registry Number | 2465-93-2 |
| EC Number | 219-573-5 |
| SMILES | C(COCC(COCCC#N)OCCC#N)C#N |
| Solubility | 6.66e+005 mg/L (25 °C water) |
|---|---|
| Density | 1.1±0.1 g/cm3, Calc.*, 1.107 g/mL |
| Index of Refraction | 1.465, Calc.* |
| Melting point | 150.16 °C |
| Boiling Point | 492.4±45.0 °C (760 mmHg), Calc.*, 410.43 °C |
| Flash Point | 200.7±21.6 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS06;GHS07 Danger Details GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H301-H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P316-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||
|
1,2,3-Tris(2-cyanoethoxy)propane, also known by its systematic name, is a chemical compound consisting of a propane backbone with three 2-cyanoethoxy groups attached at positions 1, 2, and 3. This compound is of interest primarily due to its chemical structure and the presence of cyanoethoxy functional groups, which can participate in various chemical reactions and interactions. It is synthesized by attaching cyanoethoxy groups to a propane backbone, typically through a reaction involving the appropriate precursor compounds. The discovery of 1,2,3-tris(2-cyanoethoxy)propane is tied to the development of versatile molecules with specific functional groups that can serve as intermediates in organic synthesis. These functional groups can influence the reactivity and chemical behavior of the molecule, allowing it to be used in a variety of synthetic applications. The cyanoethoxy groups provide sites for nucleophilic substitution and other types of reactivity, making this compound useful in the synthesis of more complex molecules. 1,2,3-Tris(2-cyanoethoxy)propane has been explored for its applications in materials science, particularly in the development of polymers and other macromolecular structures. The cyanoethoxy groups can interact with other functional groups in polymer chemistry, allowing for the synthesis of copolymers or the modification of existing polymeric structures. These interactions can enhance the properties of the resulting materials, such as their stability, reactivity, or solubility. Additionally, this compound has potential uses in the synthesis of specialized organic compounds. The cyanoethoxy groups can be utilized in a variety of reactions, such as nucleophilic substitution, esterification, or polymerization, allowing for the creation of new materials with specific properties. Its use in synthetic organic chemistry can also extend to the production of pharmaceutical intermediates or agrochemical compounds, where its ability to undergo chemical transformations makes it a valuable building block. The application of 1,2,3-tris(2-cyanoethoxy)propane in the field of electronic materials is another area of interest. Its molecular structure can be adapted for the development of materials with specific electronic or optical properties, such as those used in the manufacture of semiconductors, displays, or sensors. The presence of cyanoethoxy groups in the structure can influence the electronic properties of the material, making it suitable for these applications. In summary, 1,2,3-tris(2-cyanoethoxy)propane is a chemical compound with significant potential in organic synthesis and materials science. Its cyanoethoxy functional groups provide opportunities for reactivity and the development of new materials and compounds. While its specific applications may vary, its utility in the synthesis of complex molecules, polymer chemistry, and electronic materials underscores its importance in various chemical industries. References 1964. Use of 1,2,3-tris (2-cyanoethoxy)-propane as the stationary phase in gas-liquid chromatography. Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 13(1). DOI: 10.1007/bf01179597 1986. Liquid-liquid Chromatography. Practice of High Performance Liquid Chromatography. DOI: 10.1007/978-3-642-69225-3_6 2020. Gas Chromatography Columns Using Ionic Liquids as Stationary Phase. Green Chemistry and Sustainable Technology. DOI: 10.1007/978-3-030-35245-5_6 |
| Market Analysis Reports |