Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Organic raw materials >> Heterocyclic compound |
|---|---|
| Name | Acridine |
| Synonyms | 10-Azaanthracene; 2,3-Benzoquinoline; 9-Azaanthracene; Benzo[b]quinoline; Dibenzo[b,e]pyridine; NSC 3408 |
| Molecular Structure | 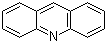 |
| Molecular Formula | C13H9N |
| Molecular Weight | 179.22 |
| CAS Registry Number | 260-94-6 |
| EC Number | 205-971-6 |
| SMILES | C1=CC=C2C(=C1)C=C3C=CC=CC3=N2 |
| Density | 1.2±0.1 g/cm3, Calc.*, 1.016 g/mL (Expl.) |
|---|---|
| Melting Point | 107-110 °C (Expl.) |
| Index of Refraction | 1.727, Calc.* |
| Boiling Point | 346.7±11.0 °C (760 mmHg), Calc.*, 346 °C (Expl.) |
| Flash Point | 153.8±11.9 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H312-H315-H319-H332-H335 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P317-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 2713 | ||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
Acridine is a heterocyclic organic compound, characterized by a fused ring structure containing nitrogen, specifically consisting of a benzene ring fused with a pyridine ring. Its chemical formula is C9H7N, and it is widely known for its yellow crystalline appearance. Acridine has been of considerable interest to chemists due to its unique structure and its potential applications across a variety of fields, including medicine, materials science, and environmental chemistry. The discovery of acridine dates back to the early 19th century when it was first isolated from coal tar by the German chemist Carl Gräbe in 1870. Gräbe's work with acridine and other compounds derived from coal tar contributed significantly to the development of organic chemistry during that era. Acridine's chemical structure was later elucidated, confirming it as a nitrogen-containing compound. Over time, it became recognized as a significant member of the class of heterocyclic aromatic compounds, which includes a variety of biologically and industrially important molecules. One of the major applications of acridine is in the field of medicine. Acridine derivatives, such as acridines and acridine orange, have been utilized as antimicrobial and anticancer agents. Acridine derivatives are known to interact with DNA, inhibiting replication and transcription processes, which makes them effective as chemotherapeutic agents in cancer treatment. Acridine orange, in particular, is widely used in molecular biology and cytogenetics as a fluorescent dye that can stain nucleic acids, allowing researchers to visualize cells and tissues under a microscope. It has applications in fluorescence microscopy and flow cytometry. Acridine is also employed in the field of materials science. As an organic compound with distinctive electronic properties, acridine and its derivatives have been explored for use in organic light-emitting diodes (OLEDs), organic solar cells, and other electronic devices. Its unique structure allows it to act as a hole-transport material, which is essential for efficient charge injection and transport in electronic devices. Furthermore, acridine-based compounds are being investigated for their potential as optoelectronic materials in the development of more efficient and flexible electronics. In environmental chemistry, acridine is utilized as an indicator for the presence of aromatic compounds in water and soil. It is used in environmental monitoring to assess contamination levels, as well as to track the biodegradation of toxic substances. The ability of acridine to bind to other organic compounds makes it a useful tool for detecting and measuring environmental pollutants. In conclusion, acridine is a versatile chemical compound with a broad range of applications in medicine, materials science, and environmental monitoring. Its discovery has paved the way for numerous advancements in drug development, electronic materials, and environmental analysis, solidifying its importance in both scientific research and industrial applications. References 2024. Nanocomposite-Based Photocatalysis: Tackling Pharmaceutical and Personal Care Products (PPCPs) Pollutants in Environmental Remediation. Sustainable Development Goals Towards Environmental Toxicity and Green Chemistry. DOI: 10.1007/978-3-031-77327-3_5 1979. Deoxyribonucleic acid bifunctional intercalators: kinetic investigation of the binding of several acridine dimers to deoxyribonucleic acid. Biochemistry. DOI: 10.1021/bi00582a023 1969. N4,N4'-Decamethylenebis(4-aminopyridine) and N9,N9'-decamethylenebis(9-aminoacridine). Journal of Medicinal Chemistry. DOI: 10.1021/jm00304a037 |
| Market Analysis Reports |