Online Database of Chemicals from Around the World
| Wuhan Kemi-works Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kemiworks.net | |||
 | +86 (27) 8573-6489 | |||
 | +86 (27) 8573-6485 | |||
 | info@kemiworks.net sales@kemiworks.com | |||
| Chemical manufacturer | ||||
| chemBlink Premium supplier since 2011 | ||||
| Classification | Organic raw materials >> Carboxylic compounds and derivatives |
|---|---|
| Name | 9-Acridinecarboxylic acid |
| Synonyms | Acridine-9-carboxylic acid |
| Molecular Structure | 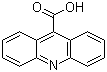 |
| Molecular Formula | C14H9NO2 |
| Molecular Weight | 223.23 |
| CAS Registry Number | 5336-90-3 |
| EC Number | 689-011-5 |
| SMILES | C1=CC=C2C(=C1)C(=C3C=CC=CC3=N2)C(=O)O |
| Density | 1.4±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 290 °C (decomp.) (Expl.) |
| Index of Refraction | 1.756, Calc.* |
| Boiling Point | 480.4±18.0 °C (760 mmHg), Calc.* |
| Flash Point | 244.3±21.2 °C, Calc.* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P270-P271-P280-P301+P317-P302+P352-P304+P340-P305+P351+P338-P319-P321-P330-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
9-Acridinecarboxylic acid is an aromatic organic compound that combines the structure of acridine with a carboxylic acid group at the 9-position. The molecular formula of 9-acridinecarboxylic acid is C9H7NO2, and it is commonly used as an intermediate in organic synthesis. The compound’s structure, which consists of a nitrogen-containing aromatic ring with a carboxyl group, contributes to its unique chemical properties, making it an important building block for the development of various chemical products. The discovery of 9-acridinecarboxylic acid traces back to the study of acridine derivatives. Acridine, a heterocyclic compound containing a nitrogen atom within its ring, has been widely explored for its applications in medicinal chemistry and material science. The introduction of a carboxylic acid group at the 9-position of the acridine ring enhances the reactivity and solubility of the molecule, opening the door for further functionalization and application in diverse chemical fields. One of the primary applications of 9-acridinecarboxylic acid is in the synthesis of fluorescent dyes and biological probes. Due to the presence of the acridine ring, which exhibits strong fluorescent properties, 9-acridinecarboxylic acid serves as a precursor for the development of compounds used in fluorescence microscopy and other analytical techniques. These derivatives are valuable tools in biochemical assays, as they can be used to label nucleic acids, proteins, or other biomolecules, allowing for the detection and study of molecular interactions in biological systems. In addition to its use in fluorescence-based applications, 9-acridinecarboxylic acid and its derivatives have been explored for their potential in medicinal chemistry. Acridine-based compounds are known to possess antitumor and antimicrobial activities, and 9-acridinecarboxylic acid derivatives have been synthesized as part of efforts to develop new chemotherapeutic agents. The carboxyl group in 9-acridinecarboxylic acid plays a key role in enhancing the compound’s solubility and bioavailability, while the acridine ring contributes to its potential to intercalate into DNA, a mechanism that is important for its anticancer properties. Moreover, 9-acridinecarboxylic acid is used in the development of materials science, specifically in the creation of conductive polymers and molecular electronics. The functionalization of the acridine core with various groups, such as carboxyl, amine, or alkyl, allows for the tailoring of the compound’s electronic properties, which can be applied to organic light-emitting diodes (OLEDs), organic photovoltaics (OPVs), and other organic electronic devices. References 2018. Synthesis of new acridine-9-carboxylic acid derivatives. Russian Chemical Bulletin, 67(5). DOI: 10.1007/s11172-018-2152-1 2020. New acridine-9-carboxamide linked to 1,2,3-triazole-N-phenylacetamide derivatives as potent α-glucosidase inhibitors: design, synthesis, in vitro, and in silico biological evaluations. Medicinal Chemistry Research, 29(10). DOI: 10.1007/s00044-020-02603-7 2005. Synthesis, Structures, and Magnetic Properties of the Copper(II), Cobalt(II), and Manganese(II) Complexes with 9-Acridinecarboxylate and 4-Quinolinecarboxylate Ligands. Inorganic Chemistry, 44(24). DOI: 10.1021/ic050886d |
| Market Analysis Reports |