Online Database of Chemicals from Around the World
| Sinocompound Catalysts Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sinocompound.com | |||
 | +86 (512) 6721-6630 | |||
 | +86 (512) 5631-6689 | |||
 | sales@sinocompound.com | |||
| Chemical manufacturer since 2008 | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Zhengzhou Kingorgchem Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.kingorgchem.com | |||
 | +86 (371) 6551-1006 | |||
 | +86 (371) 6575-6965 | |||
 | sales@kingorgchem.com | |||
 | QQ Chat | |||
 | WeChat: 18625597674 | |||
| Chemical manufacturer since 2015 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Zhengzhou Changkuan Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.changkuantech.com | |||
 | +86 (371) 6376-9919 +86 18530983798 | |||
 | +86 (371) 6360-3986 | |||
 | sales5@changkuantech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2012 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Xinxiang Runyu Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.runvmat.com | |||
 | +86 (373) 775-9608 | |||
 | +86 (373) 775-9608 | |||
 | sales@runvmat.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2017 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Dalchem | Russia | |||
|---|---|---|---|---|
 | www.dalchem.com | |||
 | +7 (8312) 753-772 | |||
 | +7 (8312) 750-799 | |||
 | dregichy@dalchem.com | |||
| Chemical manufacturer since 1997 | ||||
| Classification | Organic raw materials >> Organic phosphine compound |
|---|---|
| Name | 4,6-Bis(diphenylphosphino)phenoxazine |
| Synonyms | 4,6-Bis(diphenylphosphino)-10H-phenoxazine |
| Molecular Structure | 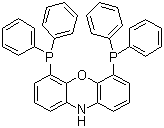 |
| Molecular Formula | C36H27NOP2 |
| Molecular Weight | 551.55 |
| CAS Registry Number | 261733-18-0 |
| EC Number | 812-116-6 |
| SMILES | C1=CC=C(C=C1)P(C2=CC=CC=C2)C3=CC=CC4=C3OC5=C(N4)C=CC=C5P(C6=CC=CC=C6)C7=CC=CC=C7 |
| Melting point | 184-186 °C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H315-H319-H335 Details | ||||||||||||||||||||
| Safety Statements | P261-P264-P264+P265-P271-P280-P302+P352-P304+P340-P305+P351+P338-P319-P321-P332+P317-P337+P317-P362+P364-P403+P233-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
4,6-Bis(diphenylphosphino)phenoxazine is a specialized organophosphorus compound known for its unique structural features and versatile applications in coordination chemistry and catalysis. This compound features a phenoxazine backbone with two diphenylphosphino groups attached at the 4 and 6 positions, which imparts significant steric and electronic properties to the molecule. The discovery of 4,6-Bis(diphenylphosphino)phenoxazine can be traced to research aimed at developing new ligands for transition metal complexes. The phenoxazine core, characterized by its fused ring system, was selected for its ability to stabilize metal centers through delocalized π-electron interactions. The introduction of diphenylphosphino groups at strategic positions enhances the ligand’s ability to engage in various coordination and catalytic processes. The synthesis of 4,6-Bis(diphenylphosphino)phenoxazine involves several key steps. The starting material is typically a phenoxazine derivative, which is then functionalized with diphenylphosphine groups. This process often begins with the preparation of the phenoxazine core through cyclization reactions involving an appropriate precursor compound. Subsequently, the diphenylphosphino groups are introduced via a phosphination reaction, where diphenylphosphine is reacted with the phenoxazine derivative under controlled conditions. The final product is purified using techniques such as column chromatography to ensure high purity and yield. One of the primary applications of 4,6-Bis(diphenylphosphino)phenoxazine is as a bidentate ligand in coordination chemistry. The ligand’s two diphenylphosphino groups can form stable chelate rings with transition metal centers, enhancing the metal’s reactivity and selectivity in various catalytic reactions. This makes it a valuable component in the development of new catalysts for a range of chemical processes, including cross-coupling reactions, hydrogenation, and oxidation. In catalytic applications, 4,6-Bis(diphenylphosphino)phenoxazine has been used to facilitate the formation of carbon-carbon and carbon-heteroatom bonds. The ligand’s ability to stabilize transition metal centers allows for efficient catalytic turnover and improved reaction yields. For instance, transition metal complexes containing this ligand are employed in Suzuki-Miyaura and Heck coupling reactions, where they aid in the formation of complex organic molecules from simpler substrates. The compound also finds use in material science, where it is utilized in the development of new materials with tailored electronic or optical properties. The phenoxazine core and diphenylphosphino groups contribute to the electronic structure of the materials, making them suitable for applications in organic light-emitting diodes (OLEDs), sensors, and other electronic devices. The advantages of using 4,6-Bis(diphenylphosphino)phenoxazine include its ability to form stable complexes with a variety of transition metals and its versatility in different catalytic processes. The ligand’s steric and electronic properties provide fine-tuning of reactivity, making it a valuable tool in both research and industrial applications. Challenges associated with this compound may include managing the stability of the metal-ligand complexes and optimizing reaction conditions for specific applications. Future research on 4,6-Bis(diphenylphosphino)phenoxazine may explore new catalytic applications, including its use in asymmetric synthesis and other advanced chemical processes. Additionally, researchers may investigate the development of new derivatives with enhanced properties for specific applications in material science and electronics. References 2008. 4,6-Bis(diphenyl-phosphino)phenoxazine (nixantphos). Acta Crystallographica. Section E, Structure Reports Online. DOI: 10.1107/s1600536808006648 2016. Recent Developments in Microwave-Assisted Metal-Catalyzed C-H Functionalization of Heteroarenes for Medicinal Chemistry and Material Applications. Synthesis. DOI: 10.1055/s-0035-1562608 2024. Highly Efficient and Linear-Selective Hydroformylation of Allyl Acetate Under Mild Conditions Regulated by Additives. Catalysis Letters. DOI: 10.1007/s10562-024-04720-z |
| Market Analysis Reports |