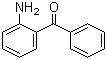
2-Aminobenzophenone, also known as o-aminobenzophenone or 2-benzoylaniline, is an aromatic ketone with the molecular formula C13H11NO. It consists of a benzophenone core in which an amino group (–NH2) is substituted at the 2-position (ortho) of one of the phenyl rings. This arrangement gives the molecule both nucleophilic and electrophilic reactive sites, enabling it to participate in a variety of chemical transformations. The compound appears as a pale yellow solid at room temperature and is moderately soluble in organic solvents such as ethanol, chloroform, and acetone.
The synthesis of 2-aminobenzophenone dates back to early developments in organic chemistry involving Friedel–Crafts acylation and nitration reactions. One traditional route to the compound involves the acylation of aniline with benzoyl chloride under acidic or basic conditions. Alternatively, benzophenone derivatives can be nitrated to yield nitrobenzophenones, which are then reduced to form the corresponding aminobenzophenones. Reduction methods commonly use tin and hydrochloric acid, catalytic hydrogenation, or iron-acid systems, and careful control is required to avoid over-reduction or side reactions.
2-Aminobenzophenone is a useful intermediate in the synthesis of heterocyclic compounds, especially those involving nitrogen-containing ring systems. One of its most well-established applications is in the synthesis of quinolines via the Friedländer synthesis, where 2-aminobenzophenone condenses with carbonyl compounds such as ketones or aldehydes under acidic or basic conditions. The resulting quinolines are important scaffolds in medicinal chemistry, with derivatives used in the development of antimalarial, antibacterial, anti-inflammatory, and anticancer agents.
Beyond quinoline synthesis, 2-aminobenzophenone also serves as a building block in the preparation of dyes, optical brighteners, and agrochemical compounds. The amino group can undergo diazotization followed by coupling with electron-rich aromatic systems to form azo dyes, which are used for coloring textiles and polymers. Additionally, the ketone functionality enables participation in various condensation reactions to form Schiff bases, hydrazones, or enamine derivatives, which may exhibit coordination behavior or biological activity.
The compound has been investigated for its spectroscopic properties, and both the amino and carbonyl functional groups contribute to its ultraviolet (UV) absorbance. This characteristic makes it useful as a standard or probe in analytical studies involving UV-visible spectroscopy. Furthermore, its fluorescence properties, though relatively weak, have also been studied in relation to solvent polarity and substituent effects on the benzophenone ring system.
From a safety standpoint, 2-aminobenzophenone should be handled with caution. Aminoaromatic compounds may cause skin and respiratory irritation and should be managed in a well-ventilated area using appropriate personal protective equipment. Although 2-aminobenzophenone is not classified as highly hazardous, exposure should be minimized, and all standard laboratory safety procedures should be observed. Waste materials should be disposed of in accordance with local environmental and chemical safety regulations.
In summary, 2-aminobenzophenone is a synthetically versatile aromatic compound that plays an important role in organic chemistry. Its dual functionality—amino and ketone groups—makes it a valuable intermediate in the synthesis of heterocycles, dyes, and bioactive compounds. Its historical and continuing significance lies in its utility as a starting material for quinoline derivatives and related compounds with diverse industrial and pharmaceutical applications.
References
2023. Current progress toward synthetic routes and medicinal significance of quinoline. Medicinal Chemistry Research, 32(9).
DOI: 10.1007/s00044-023-03121-y
2022. Microwave irradiation for the synthesis of quinoline scaffolds: a review. Journal of the Iranian Chemical Society, 19(9).
DOI: 10.1007/s13738-022-02648-y
2022. Visible-light-driven radical Friedländer hetero-annulation of 2-aminoaryl ketone and α-methylene carbonyl compound via organic dye fluorescein through a single-electron transfer (SET) pathway. BMC Chemistry, 16(1).
DOI: 10.1186/s13065-022-00910-1
|





































































 GHS07 Warning Details
GHS07 Warning Details