Online Database of Chemicals from Around the World
| Tianjin Zhongxin Chem-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tjzxchem.com | |||
 | +86 (22) 6688-0623 | |||
 | +86 (22) 8988-0739 ex 8030 | |||
 | sales@tjzxchem.com | |||
| Chemical manufacturer since 2007 | ||||
| chemBlink Premium supplier since 2009 | ||||
| Classification | Chemical reagent >> Organic reagent >> Amide |
|---|---|
| Name | Diacetoneacrylamide |
| Synonyms | 4-Acrylamido-4-methyl-2-pentanone; Diacetone acrylamide; N-(1,1-Dimethyl-3-oxobutyl)acrylamide |
| Molecular Structure | 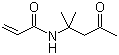 |
| Molecular Formula | C9H15NO2 |
| Molecular Weight | 169.22 |
| CAS Registry Number | 2873-97-4 |
| EC Number | 220-713-2 |
| SMILES | CC(=O)CC(C)(C)NC(=O)C=C |
| Density | 1.0±0.1 g/cm3, Calc.* |
|---|---|
| Melting point | 53-57 °C |
| Index of Refraction | 1.450, Calc.* |
| Boiling Point | 325.9±34.0 °C (760 mmHg), Calc.*, 120 °C (8 mmHg) |
| Flash Point | 139.4±25.8 °C, Calc.*, 110 °C |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS07;GHS08 Warning Details GHS07;GHS08 Warning Details | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H361 Details | ||||||||||||
| Safety Statements | P203-P264-P270-P280-P301+P317-P318-P330-P405-P501 Details | ||||||||||||
| Hazard Classification | |||||||||||||
| |||||||||||||
| SDS | Available | ||||||||||||
|
Diacetoneacrylamide (DAAM) is an important organic compound that has garnered significant attention in the field of polymer chemistry. It is classified as an acrylamide derivative, characterized by the presence of both a carbonyl group and a vinyl group, which enables it to undergo polymerization. DAAM's unique structure contributes to its reactivity, making it a versatile monomer used in various applications, particularly in the synthesis of polymers and copolymers. The discovery of diacetoneacrylamide can be traced back to the early exploration of acrylamide derivatives in the mid-20th century. Researchers aimed to develop new monomers with enhanced properties for use in industrial applications. DAAM was synthesized through the reaction of diacetone alcohol and acrylonitrile, a process that highlights its versatility as a building block in organic synthesis. The resulting compound exhibits a range of properties that make it suitable for various applications, particularly in the formulation of adhesives, coatings, and hydrogels. One of the primary applications of diacetoneacrylamide is in the production of water-soluble polymers. When polymerized, DAAM forms a highly flexible and transparent polymer that is soluble in water, making it suitable for various formulations. Its ability to create hydrophilic polymers has made it a popular choice in the development of hydrogels, which are used in a wide range of applications, including drug delivery systems, wound dressings, and contact lenses. The hydrophilic nature of DAAM-based polymers allows them to absorb large amounts of water, which is critical for maintaining moisture in medical applications. In addition to its use in hydrogels, diacetoneacrylamide is also utilized in the formulation of coatings and adhesives. The compound's unique properties enable it to enhance adhesion and improve the durability of coatings, making it suitable for various industrial applications. DAAM-based coatings exhibit excellent chemical resistance, making them ideal for use in automotive, aerospace, and construction industries, where performance and longevity are crucial. Moreover, diacetoneacrylamide has gained attention in the field of biomaterials. Its biocompatibility and ability to form hydrogels make it an attractive candidate for use in tissue engineering and regenerative medicine. Researchers have explored the potential of DAAM-based hydrogels as scaffolds for cell growth and tissue repair, highlighting the compound's versatility in biomedical applications. Despite its numerous applications, the use of diacetoneacrylamide is accompanied by certain safety considerations. Like other acrylamide derivatives, DAAM can pose health risks if handled improperly. It is important for manufacturers and researchers to follow appropriate safety protocols to minimize exposure and ensure safe handling during production and application. Ongoing research into diacetoneacrylamide aims to further explore its potential applications and enhance its properties. Innovations in polymer chemistry are focused on optimizing the synthesis of DAAM-based materials, as well as developing new formulations that leverage its unique characteristics. The versatility of diacetoneacrylamide positions it as a valuable compound in the fields of materials science and biomedical engineering. The discovery and application of diacetoneacrylamide illustrate the significance of acrylamide derivatives in advancing chemical innovation. Its roles in hydrogels, coatings, and biomaterials underscore the impact of DAAM in addressing contemporary challenges across various sectors. References 2024. Advancements and applications of dosimetry techniques in modern medical radiation therapy: a comprehensive review. Journal of Radioanalytical and Nuclear Chemistry. DOI: 10.1007/s10967-024-09517-3 2024. Rheology Analysis of Molecular Structures of Modified HPAM Polymers on Heat Resistance. Proceedings of the International Field Exploration and Development Conference 2023. DOI: 10.1007/978-981-97-0264-0_70 2023. Preparation of Pickering emulsion hydrogels containing indium(III) extractants and their indium(III) recycling properties. Clean Technologies and Environmental Policy. DOI: 10.1007/s10098-023-02545-9 |
| Market Analysis Reports |